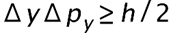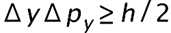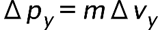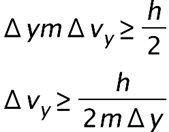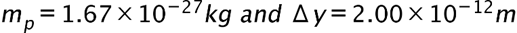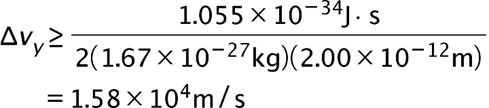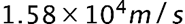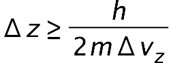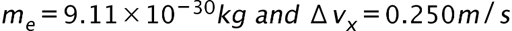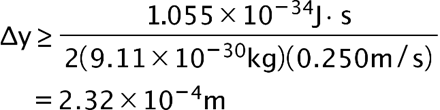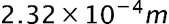Chapter 6: Q45E (page 1315)
(a) The uncertainty in the y-component of a proton’s position is. What is the minimum uncertainty in a simultaneous measurement of the y-component of the proton’s velocity? (b) The uncertainty in the z-component of an electron’s velocity is 0.250 m/s. What is the minimum uncertainty in a simultaneous measurement of the z-coordinate of the electron?
Short Answer
- The minimum uncertainty in the proton's y-component speed is
- The minimum uncertainty in the proton’s z-component position is