Chapter 1: Q59. (page 20)
One mole of atoms consists of individual atoms. If a mole of atoms were spread uniformly over the Earth’s surface, how many atoms would there be per square meter?
Short Answer
There would be over the surface of the Earth.
Learning Materials
Features
Discover
Chapter 1: Q59. (page 20)
One mole of atoms consists of individual atoms. If a mole of atoms were spread uniformly over the Earth’s surface, how many atoms would there be per square meter?
There would be over the surface of the Earth.
All the tools & learning materials you need for study success - in one app.
Get started for free
Express the following using the prefixes of Table 1–4:
(a)
(b)
(c)
(d)
(e)
What, approximately, is the percent uncertainty for a measurement given as?
Time intervals measured with a stopwatch typically have an uncertainty of about 0.2 s due to human reaction time at the start and stop moments. What is the percent uncertainty of a hand-timed measurement of (a) 5.5 s, (b) 55 s, (c) 5.5 min?
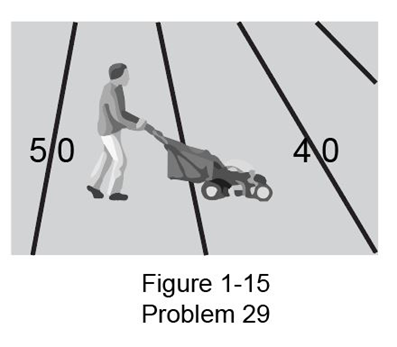
Estimate how long it would take one person to mow a football field using an ordinary home lawnmower (Fig. 1-15). (State your assumption, such as the mower moves with a 1 km/h speed and has a 0.5 m width.)
(Note: Remember that for rough estimates, only round numbers are needed both as input to calculations and as final results.)
Express the sine ofwith the correct number of significant figures.
What do you think about this solution?
We value your feedback to improve our textbook solutions.