Chapter 1: Q59E (page 1)
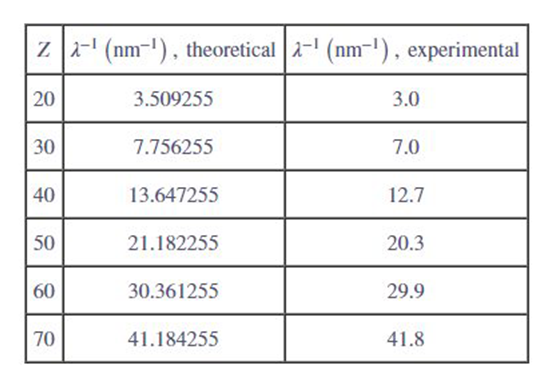
Estimate characteristic X-ray wavelengths: A hole has already been produced in the n=1 shell, and an n=2 electron is poised to jump in. Assume that the electron behaves as though in a "hydrogenlike" atom (see Section 7.8), with energy given by . Before the jump, it orbits Z protons, one remaining electron. and (on average) half its seven fellow n=2 electrons, for a of . After the jump, it orbits Z protons and half of its fellow electron, for a of . Obtain a formula for versus Z . Compare the predictions of this model with the experimental data given in Figure and Table .
Short Answer
The formula for versus Z is and on comparison, the theoretical and experimental values are reasonably close.