Chapter 9: Q60E (page 407)
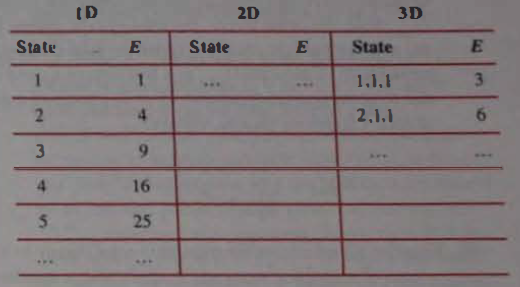
For a particle in a one-dimensional (ID) box, is proportional to a single quantum number . Let us simplify things by ignoring the proportionality factor: . For a 3D box, , and the 2D box is fairly obvious.
(a) The table shows a start on accounting for allowed states. Complete the table, stopping after the 10th state (state, not energy) for all three cases.
(b) Find the number of states per energy difference for the first five states and the last five states for all three cases. For instance, for the first five in the ID case, it is 5 states per energy difference of 24, or .
(c) Overlooking the obviously crude aspects of this accounting, does the "density of states" seem to increase with energy, decrease with energy, or stay about the same?
Short Answer
(a). Complete table is given in the solution
(b). The number of states for first five and last five inareandrespectively, in 2D areandrespectively and in 3D areandrespectively.
c). The density of the states tends to decrease with the energy.

