Chapter 9: Q27E (page 404)
Four distinguishable Hamonic oscillators may exchange energy. The energies allowed particleare: those allowed particlebare, and so on. Consider an overall state (macro-state) in which the total energy is. One possible microstate would have particles in theirstates and particle d in itsstates that is,.
(a) List all possible microstates, (b) What is the probability that a given particle will be in its state? (c) Answer part (b) for all other possible values of n. (d) Plot the probability versus n.
Short Answer
- The list of all possible microstates, for the given microstate, is given below-
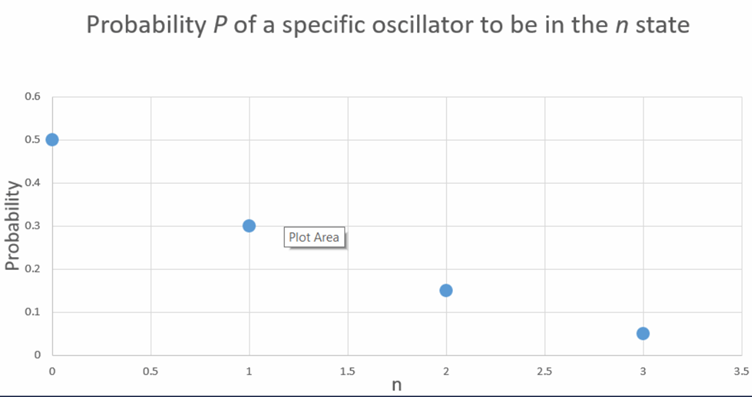
- Probability of a particle to be in state is.
- The probability for particle to in, , and are respectively.
- The plot for probability vs n is shown in part (d) below.