Chapter 8: Q50E (page 342)
Determine the expected valence of the element with atomic number 117.
Short Answer
The expected valence of the element with atomic numberis or .
Learning Materials
Features
Discover
Chapter 8: Q50E (page 342)
Determine the expected valence of the element with atomic number 117.
The expected valence of the element with atomic numberis or .
All the tools & learning materials you need for study success - in one app.
Get started for free
In its ground state, nitrogen's 2p electrons interact to produce . Given Hund's rule, how might the orbit at angular momenta of these three electrons combine?
A hydrogen atom in its ground state is subjected to an external magnetic field of 1.0 T. What is the energy difference between the spin-up and spin-down states?
The Zeeman effect occurs in sodium just as in hydrogen-sodium's lone valence electron behaves much as hydrogen's 1.5. Suppose sodium atoms are immersed in amagnetic field.
(a) Into how many levels is thelevel split?
(b) Determine the energy spacing between these states.
(c) Into how many lines is thetospectral line split by the field?
(d) Describe quantitatively the spacing of these lines.
(e) The sodium doublet is two spectral lines.and. which are split according to the two differentpossible spin-orbit energies in the 3Pstate (see Exercise 60). Determine the splitting of the sodium doublet (the energy difference between the two photons). How does it compare with the line splitting of part (d), and why?
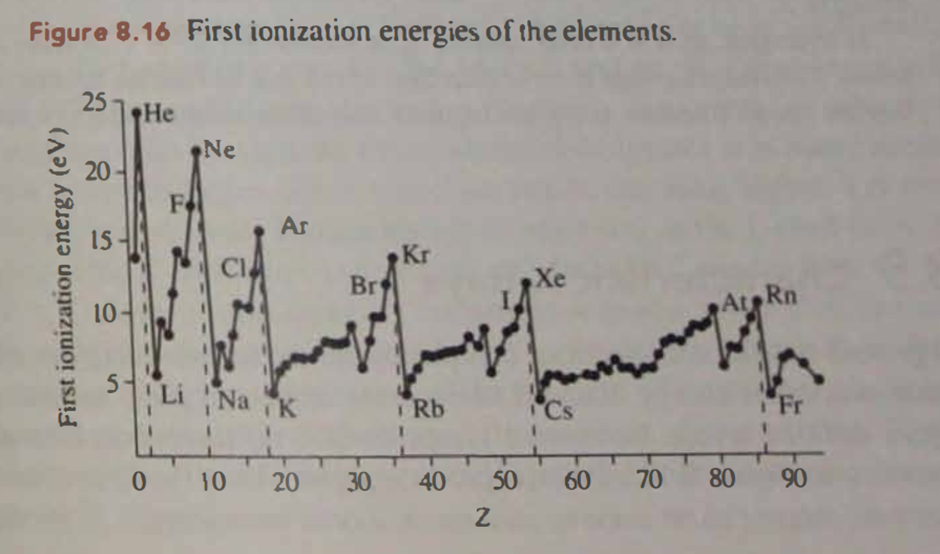
Question: Early on, the lanthanides were found to be quite uncooperative when attempts were made to chemically separate them from one another. One reason can be seen in Figure 8.16. Explain.
Question: Bearing in mind its limiting cases of 1 and 2 mentioned in section 8.8, how would you describe the significance of the Lande g-factor
What do you think about this solution?
We value your feedback to improve our textbook solutions.