Chapter 5: Q83CE (page 193)
Calculate the uncertainty in the particle’s position.
Short Answer
The uncertainty in the particle position is .
Learning Materials
Features
Discover
Chapter 5: Q83CE (page 193)
Calculate the uncertainty in the particle’s position.
The uncertainty in the particle position is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Simple models are very useful. Consider the twin finite wells shown in the figure, at First with a tiny separation. Then with increasingly distant separations, In all case, the four lowest allowed wave functions are planned on axes proportional to their energies. We see that they pass through the classically forbidden region between the wells, and we also see a trend. When the wells are very close, the four functions and energies are what we might expect of a single finite well, but as they move apart, pairs of functions converge to intermediate energies.
(a) The energies of the second and fourth states decrease. Based on changing wavelength alone, argue that is reasonable.
(b) The energies of the first and third states increase. Why? (Hint: Study bow the behaviour required in the classically forbidden region affects these two relative to the others.)
(c) The distant wells case might represent two distant atoms. If each atom had one electron, what advantage is there in bringing the atoms closer to form a molecule? (Note: Two electrons can have the same wave function.)

does the wave function have a well-defined momentum? Explain.
An electron in the n=4 state of a 5 nm wide infinite well makes a transition to the ground state, giving off energy in the form of photon. What is the photon’s wavelength?
Verify that solution (5-19) satisfies the Schrodinger equation in form (5.18).
The figure shows a potential energy function.
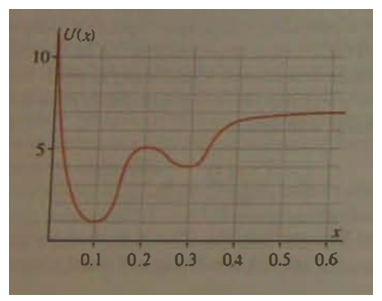
(a) How much energy could a classical particle have and still be bound?
(b) Where would an unbound particle have its maximum kinetic energy?
(c) For what range of energies might a classical particle be bound in either of two different regions?
(d) Do you think that a quantum mechanical particle with energy in the range referred to in part?
(e) Would be bound in one region or the other? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.