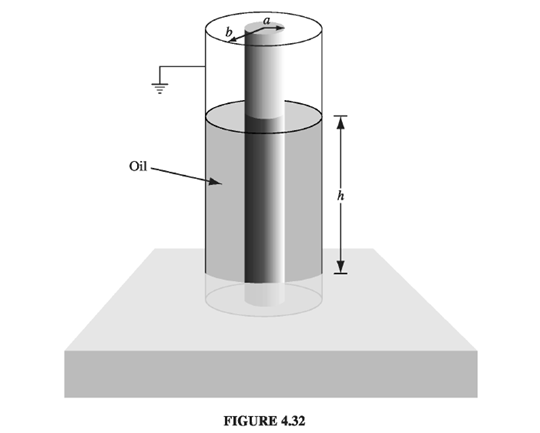
Chapter 4: Q4.42P (page 209)
Check the Clausius-Mossotti relation (Eq. 4.72) for the gases listed in Table 4.1. (Dielectric constants are given in Table 4.2.) (The densities here are so small that Eqs. 4.70 and 4.72 are indistinguishable. For experimental data that confirm the Clausius-Mossotti correction term see, for instance, the first edition of Purcell's Electricity and Magnetism, Problem 9.28.)
Short Answer
The susceptibilities of gases in 4.1 and susceptibilities of gases in 4.2 are good agreement.