Chapter 19: Q81P (page 581)
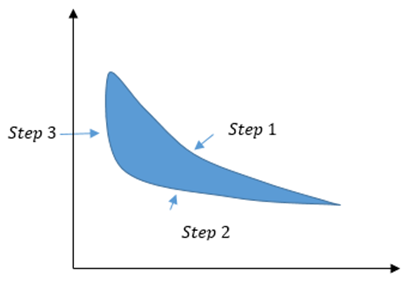
An ideal gas is taken through a complete cycle in three steps: adiabatic expansion with work equal to, isothermal contraction at, and increase in pressure at constant volume.
(a) Draw a p-V diagram for the three steps
. (b) How much energy is transferred as heat in step3, and
(c) is it transferred to or from the gas?
Short Answer
- P-V diagram is shown below.
- The work done in step 3 of the process is .
- As the internal energy of the gas is increasing, so, the heat energy is transferred to the gas.