Chapter 19: Q64P (page 581)
Calculate the work done by an external agent during an isothermal compression ofof oxygen from a volume ofatandto a volume of.
Short Answer
The work done by an external agent during the isothermal compression is.
Learning Materials
Features
Discover
Chapter 19: Q64P (page 581)
Calculate the work done by an external agent during an isothermal compression ofof oxygen from a volume ofatandto a volume of.
The work done by an external agent during the isothermal compression is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the average value of the translational kinetic energy of the molecules of an ideal gas at
a.
b.
What is the translational kinetic energy per mole of an ideal gas at
c.
d.
The dot in Fig represents the initial state of a gas, and the adiabatic through the dot divides the p-V diagram into regions and . For the following processes, determine whether the corresponding heat is positive, negative, or zero: (a) the gas moves up along the adiabatic, (b) it moves down along the adiabatic, (c) it moves to anywhere in region , and (d) it moves to anywhere in region .
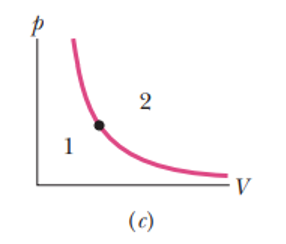
Figure shows a hypothetical speed distribution for a sample of N gas particles (note that for speed , ).
a) What is?
b) What is?
c) What is?
d) What fraction of the particles has a speed betweenand?
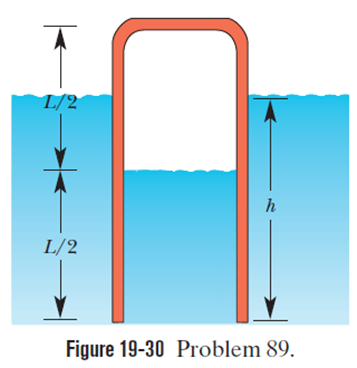
Question: A pipe of length L = 25. 0 m that is open at one end contains air at atmospheric pressure. It is thrust vertically into a freshwater lake until the water rises halfway up in the pipe (Fig.). What is the depth h of the lower end of the pipe? Assume that the temperature is the same everywhere and does not change.
A hydrogen molecule (diameter ), travelling at the rms speed, escapes from a furnace into a chamber containing argon atoms (diameter) at a density of.
a) What is the speed of the hydrogen molecule?
b) If it collides with an argon atom, what is the closest their centres can be, considering each as spherical?
c) What is the initial number of collisions per second experienced by the hydrogen molecule? (Hint: Assume that the argon atoms are stationary. Then the mean free path of the hydrogen molecule is given by
Mean free path
What do you think about this solution?
We value your feedback to improve our textbook solutions.