Chapter 18: Q9P (page 541)
A circular hole in an aluminum plate isin diameter at .What is its diameter when the temperature of the plate is raised to?
Short Answer
The diameter of the circular hole when the temperature is raised is
Learning Materials
Features
Discover
Chapter 18: Q9P (page 541)
A circular hole in an aluminum plate isin diameter at .What is its diameter when the temperature of the plate is raised to?
The diameter of the circular hole when the temperature is raised is
All the tools & learning materials you need for study success - in one app.
Get started for free
In a certain experiment, a small radioactive source must move at selected, extremely slow speeds. This motion is accomplished by fastening the source to one end of an aluminum rod and heating the central section of the rod in a controlled way. If the effective heated section of the rod in Figure has length , at what constant rate must the temperature of the rod be changed if the source is to move at a constant speed of ?
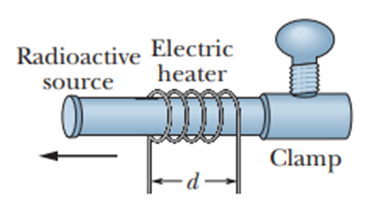
Figure 18-49 shows (in cross section) a wall consisting of four layers, with thermal conductivities , , and ( is not known). The layer thicknesses are , , and ( is not known). The known temperatures are , ,and . Energy transfer through the wall is steady. What is interface temperature ?
An aluminum-alloy rod has a length of 10.00 cmat 20.000Cand a length of 10.015 cmat the boiling point of water.
(a) What is the length of the rod at the freezing point of water?
(b) What is the temperature if the length of the rod is 10.009 cm?
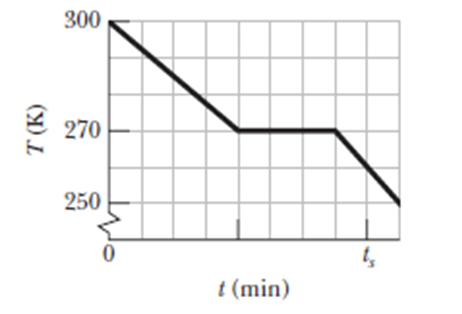
A 0.400 kg sample is placed in a cooling apparatus that removes energy as heat at a constant rate. Figure 18-32 gives the temperature T of the sample versus time t; the horizontal scale is set by ts=80.0 min. The sample freezes during the energy removal. The specific heat of the sample in its initial liquid phase is 300 J/kgK . (a) What is the sample’s heat of fusion and (b) What is its specific heat in the frozen phase?
ABuick moving atbrakes to a stop, at uniform deceleration and without skidding, over a distance of. At what average rate is mechanical energy transferred to thermal energy in the brake system?
What do you think about this solution?
We value your feedback to improve our textbook solutions.