Chapter 18: Q27P (page 542)
Calculate the minimum amount of energy, in joules, required to completely meltof silver initially at.
Short Answer
The minimum amount of energy required to completely melt 130 g of silver is
Learning Materials
Features
Discover
Chapter 18: Q27P (page 542)
Calculate the minimum amount of energy, in joules, required to completely meltof silver initially at.
The minimum amount of energy required to completely melt 130 g of silver is
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Suppose that on a linear temperature scale X, water boils at and freezes at. What is a temperature ofon the X scale? (Approximate water’s boiling point as 373K.)
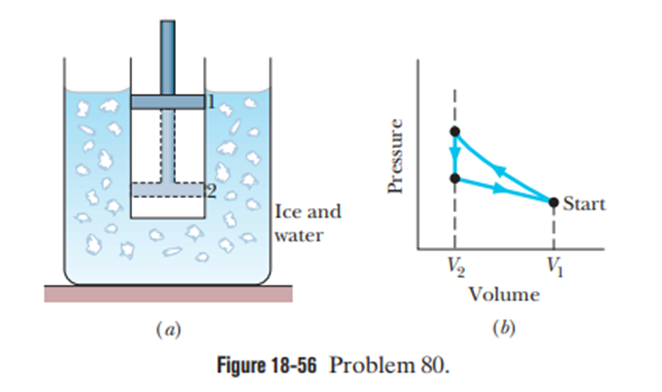
Figure 18-56ashows a cylinder containing gas and closed by a movable piston.The cylinder is kept submerged in an ice–water mixture. The piston is quicklypushed down from position 1 to position 2 and then held at position 2 until the gas is again at the temperature of the ice–water mixture; it then is slowlyraised back to position 1. Figure 18-56bis a p-Vdiagram for the process. Ifof ice is melted during the cycle, how much work has been done onthe gas?
Icebergs in the North Atlantic present hazards to shipping, causing the lengths of shipping routes to be increased by aboutduring the iceberg season. Attempts to destroy icebergs include planting explosives, bombing, torpedoing, shelling, ramming, and coating with black soot. Suppose that direct melting of the iceberg, by placing heat sources in the ice, is tried. How much energy as heat is required to melt of an iceberg that has a mass of ? (Use )
Question: (a) In, the temperature in the Siberian village of Oymyakon reached..What temperature is this on the Fahrenheit scale?
(b) The highest officially recorded temperature in the continental United States wasin Death Valley, California. What is this temperature on the Celsius scale?
Non-metric version: (a) how long does a water heater take to raise the temperature of data-custom-editor="chemistry" of water from role="math" localid="1662377531189" ? Metric version :(b) How long does a 59 kWwater heater take to raise the temperature of 150 L of water from role="math" localid="1662377622926" ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.