Chapter 42: Q9P (page 1307)
Find the disintegration energy Q for the decay of by Kelectron capture (see Problem 54). The needed data are and .
Short Answer
The disintegration energy for the decay of vanadium atom is 600 keV .
Learning Materials
Features
Discover
Chapter 42: Q9P (page 1307)
Find the disintegration energy Q for the decay of by Kelectron capture (see Problem 54). The needed data are and .
The disintegration energy for the decay of vanadium atom is 600 keV .
All the tools & learning materials you need for study success - in one app.
Get started for free
The magic nucleon numbers for nuclei are given in Module 42-8 as 2, 8, 20, 28, 50, 82, and 126. Are nuclides magic (that is, especially stable) when (a) only the mass number A, (b) only the atomic number Z, (c) only the neutron number N, or (d) either Zor N(or both) is equal to one of these numbers? Pick all correct phrases.
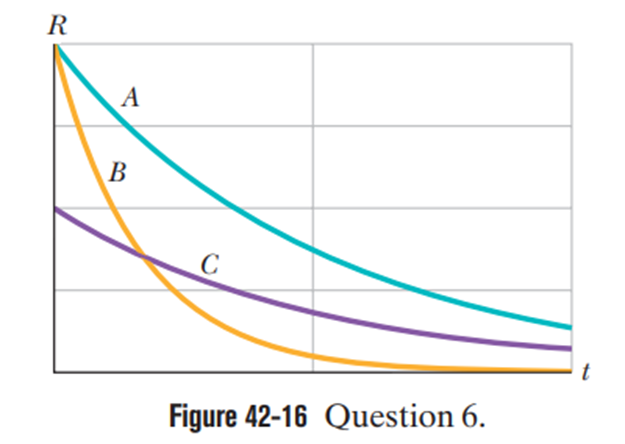
Figure 42-16 gives the activities of three radioactive samples versus time. Rank the samples according to their (a) half-life and (b) disintegration constant, greatest first. (Hint:For (a), use a straightedge on the graph.)
Two radioactive materials that alpha decay,and , and one that beta decays, are sufficiently abundant in granite to contribute significantly to the heating of Earth through the decay energy produced. The alpha-decay isotopes give rise to decay chains that stop when stable lead isotopes are formed. The isotopehas single beta decay. (Assume this is the only possible decay of that isotope.) Here is the information:
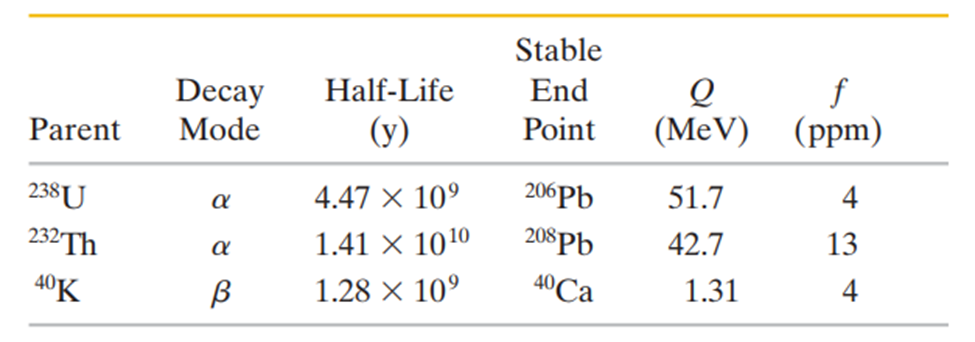
In the table Qis the totalenergy released in the decay of one parent nucleus to the finalstable endpoint and fis the abundance of the isotope in kilograms per kilogram of granite;means parts per million. (a) Show that these materials produce energy as heat at the rate offor each kilogram of granite. (b) Assuming that there isof granite in a 20-km-thick spherical shell at the surface of Earth, estimate the power of this decay process over all of Earth. Compare this power with the total solar power intercepted by Earth,1.
A rock recovered from far underground is found to contain 0.86 mg of , 0.15 mg of , and 1.6 mg of . How much will it likely contain? Assume that decays to only with a half-life of . Also assume that has a half-life of .
Make a nuclidic chart similar to Fig. 42-6 for the 25 nuclides , , , , and . Draw in and label (a) all isobaric (constant A) lines and (b) all lines of constant neutron excess, defined as N - Z.
What do you think about this solution?
We value your feedback to improve our textbook solutions.