Chapter 42: Q82P (page 1307)
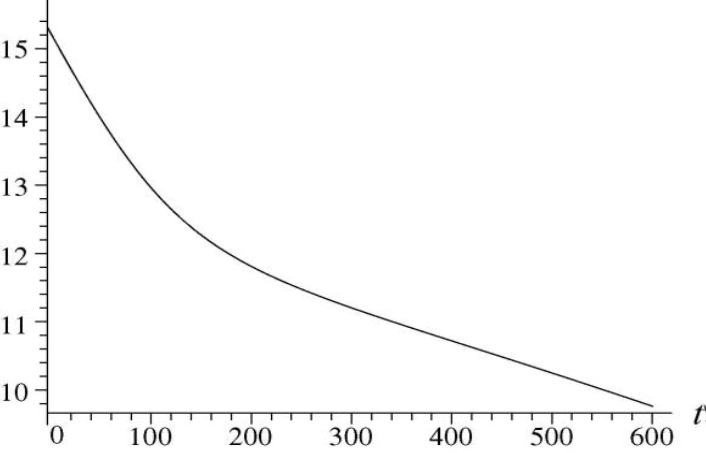
After a brief neutron irradiation of silver, two isotopes are present: with an initial decay rate of ,and role="math" localid="1661598035621" with an initial decay rate of. Make a semilog plot similar to Fig. 42-9 showing the total combined decay rate of the two isotopes as a function of time t = 0 from until t = 10min .We used Fig. 42-9 to illustrate the extraction of the half-life for simple (one isotope) decays. Given only your plot of total decay rate for the two-isotope system here, suggest a way to analyze it in order to find the half-lives of both isotopes.
Short Answer
The plot similar to the total combined decay rate of the two isotopes as a function of time from t = 0 to t = 10min is plotted.