Chapter 39: Q58P (page 1217)
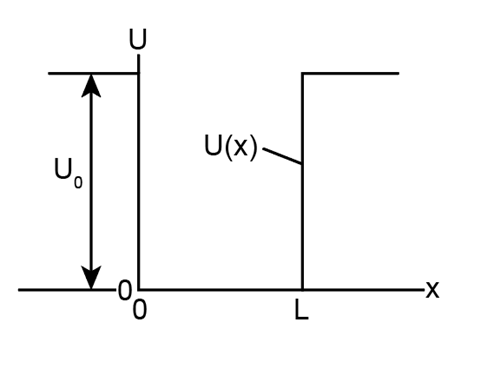
As Fig. 39-8 suggests, the probability density for the region
0 < x < L for the finite potential well of Fig. 39-7 is sinusoidal, being given by
, in which B is a constant. (a) Show that the wave function
may be found from this equation is a solution of Schrodinger’s equation in its one-dimensional form. (b) Express an equation for that makes this true.
Short Answer
(a) The proposed function satisfies Schrodinger’s equation.
(b)