Chapter 39: Q15Q (page 1214)
Identify the correspondence principle.
Short Answer
For a large system, the correspondence principles are identified, where calculations of Quantum and Classical physics match.
Learning Materials
Features
Discover
Chapter 39: Q15Q (page 1214)
Identify the correspondence principle.
For a large system, the correspondence principles are identified, where calculations of Quantum and Classical physics match.
All the tools & learning materials you need for study success - in one app.
Get started for free
A hydrogen atom can be considered as having a central point- like proton of positive charge eand an electron of negative charge -ethat is distributed about the proton according to the volume charge density. Hereis a constant,, andris the distance from the center of the atom.
(a) Using the fact that the hydrogen is electrically neutral, find A. the
(b) Then find magnitude
(c) Then find direction of the atom’s electric field at.
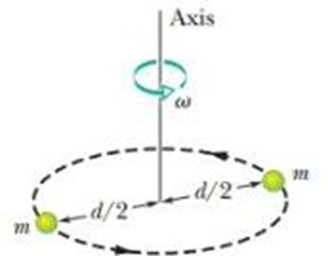
A diatomic gas molecule consists of two atoms of massseparated by a fixed distance drotating about an axis as indicated in given figure. Assuming that its angular momentum is quantized as in the Bohr model for the hydrogen atom, find
A proton is confined to a one-dimensional infinite potential well 100pm wide. What is its ground-state energy?
Consider a conduction electron in a cubical crystal of a conducting material. Such an electron is free to move throughout the volume of the crystal but cannot escape to the outside. It is trapped in a three-dimensional infinite well. The electron can move in three dimensions so that its total energy is given by
in whichare positive integer values. Calculate the energies of the lowest five distinct states for a conduction electron moving in a cubical crystal of edge length .
Calculate the energy change required for an electron to move between states: a quantum jump up or down an energy-level diagram.
What do you think about this solution?
We value your feedback to improve our textbook solutions.