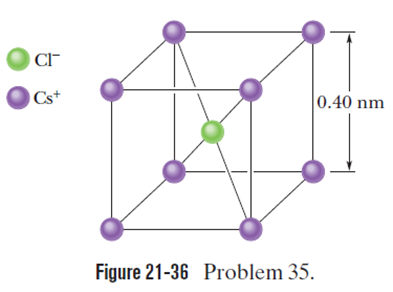
Chapter 21: Q35P (page 627)
In crystals of the salt cesium chloride, cesium ions form the eight corners of a cube and a chlorine ion is at the cube’s center (Fig. 21-36). The edge length of the cube is . The ions are each deficient by one electron (and thus each has a charge of role="math" localid="1661790179659" ), and the ion has one excess electron (and thus has a charge of ). (a)What is the magnitude of the net electrostatic force exerted on the ion by the eight ions at the corners of the cube? (b) If one of theions is missing, the crystal is said to have a defect; what is the magnitude of the net electrostatic force exerted on theion by the seven remainingions?
Short Answer
- The magnitude of the net electrostatic force exerted on the chlorine ion by the eight cesium ions at the corners of the cube is zero.
- The magnitude of the net electrostatic force exerted on the chlorine ion by the seven cesium ions at the corners of the cube is .