Chapter 40: Q10Q (page 1246)
Consider the elements krypton and Rubidium.
(a) Which is more suitable for use in a Stern–Gerlach experiment of the kind described in connection with Fig. 40-8?
(b) Which, if either, would not work at all?
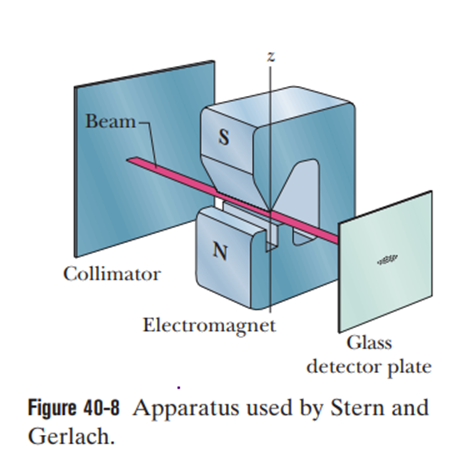
Short Answer
a) The element Rubidium is more suitable for the Stern-Gerlach experiment.
b) If both would not work at all, then an element with neutral electrical properties would be suitable for the experiment.


