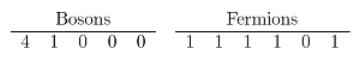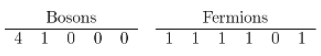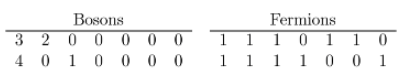Chapter 7: Q. 7.10 (page 265)
Consider a system of five particles, inside a container where the allowed energy levels are nondegenerate and evenly spaced. For instance, the particles could be trapped in a one-dimensional harmonic oscillator potential. In this problem you will consider the allowed states for this system, depending on whether the particles are identical fermions, identical bosons, or distinguishable particles.
(a) Describe the ground state of this system, for each of these three cases.
(b) Suppose that the system has one unit of energy (above the ground state). Describe the allowed states of the system, for each of the three cases. How many possible system states are there in each case?
(c) Repeat part (b) for two units of energy and for three units of energy.
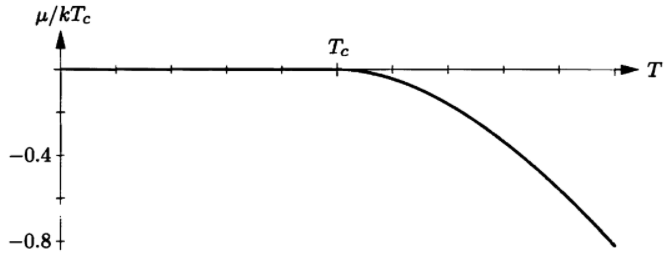
(d) Suppose that the temperature of this system is low, so that the total energy is low (though not necessarily zero). In what way will the behavior of the bosonic system differ from that of the system of distinguishable particles? Discuss.
Short Answer
(a) The ground state of this system is .
.
(b) The are five possible system states in each case.
(c) For two-unit of energy the graph is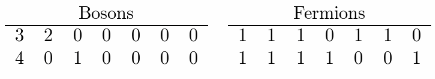
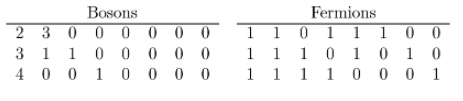
and for three-unit of energy is
 .
.
(d) The way that the behavior of the bosonic system differs from that of the system of distinguishable particles is discussed below.