Chapter 5: Q 5.31 (page 172)
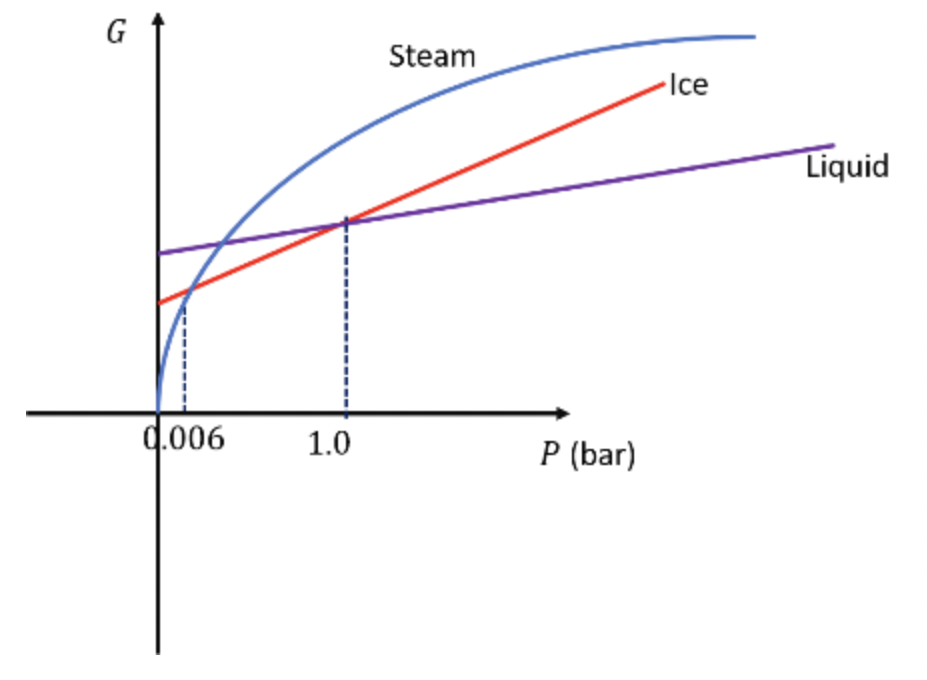
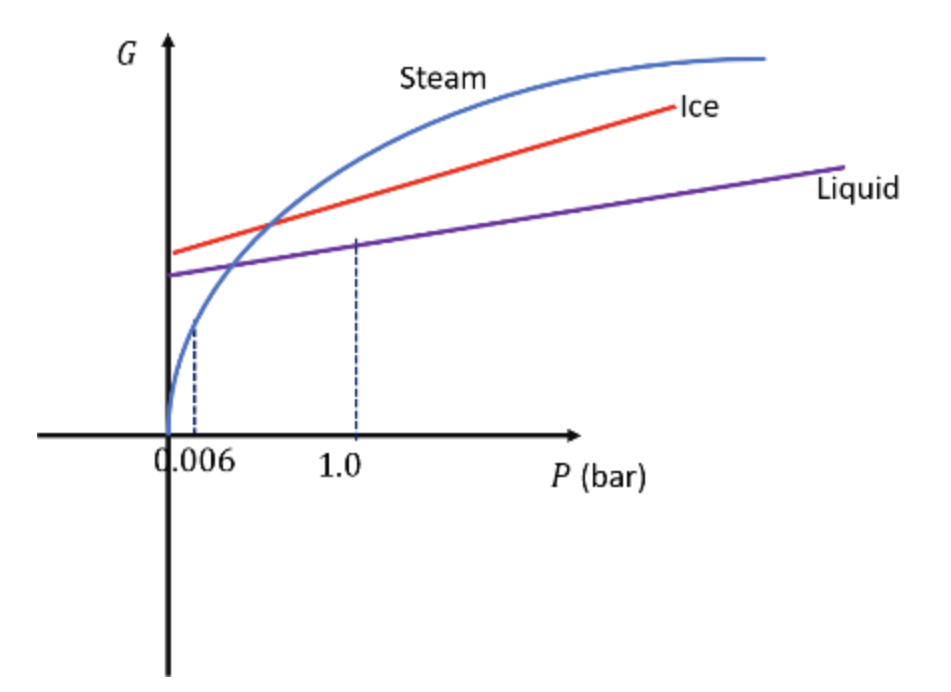
Sketch qualitatively accurate graphs of G vs. P for the three phases of H20 (ice, water, and steam) at 0°C. Put all three graphs on the same set of axes, and label the point corresponding to atmospheric pressure. How would |the graphs differ at slightly higher temperatures?
Short Answer
As the temperature rises above 0°C, the ice curve shifts to the left of the steam curve.