Chapter 4: 4.7 (page 129)
Why must you put an air conditioner in the window of a building, rather than in the middle of a room?
Short Answer
We must put t an air conditioner in the window to cool the room effectively.
Learning Materials
Features
Discover
Chapter 4: 4.7 (page 129)
Why must you put an air conditioner in the window of a building, rather than in the middle of a room?
We must put t an air conditioner in the window to cool the room effectively.
All the tools & learning materials you need for study success - in one app.
Get started for free
The magnetic field created by a dipole has a strength of approximately , where r is the distance from the dipole and is the "permeability of free space," equal to exactly in SI units. (In the formula I'm neglecting the variation of field strength with angle, which is at most a factor of 2.) Consider a paramagnetic salt like iron ammonium alum, in which the magnetic moment of each dipole is approximately one Bohr magneton , with the dipoles separated by a distance of . Assume that the dipoles interact only via ordinary magnetic forces.
(a) Estimate the strength of the magnetic field at the location of a dipole, due to its neighboring dipoles. This is the effective field strength even when there is no externally applied field.
(b) If a magnetic cooling experiment using this material begins with an external field strength of , by about what factor will the temperature decrease when the external field is turned off?
(c) Estimate the temperature at which the entropy of this material rises most steeply as a function of temperature, in the absence of an externally applied field.
(d) If the final temperature in a cooling experiment is significantly less than the temperature you found in part (c), the material ends up in a state where is very small and therefore its heat capacity is very small. Explain why it would be impractical to try to reach such a low temperature with this material.
Liquid HFC-134a at its boiling point at 12 bars pressure is throttled to 1 bar pressure. What is the final temperature? What fraction of the liquid vaporizes?
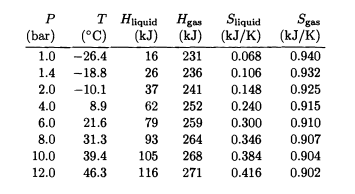
Table 4.3. Properties of the refrigerant HFC-134a under saturated conditions (at its boiling point for each pressure). All values are for of fluid, and are measured relative to an arbitrarily chosen reference state, the saturated liquid at c. Excerpted from Moran and Shapiro (1995).
Calculate the efficiency of a Rankine cycle that is modified from the parameters used in the text in each of the following three ways (one at a time), and comment briefly on the results:
(a) reduce the maximum temperature to 500oC;
(b) reduce the maximum pressure to 100 bars;
(c) reduce the minimum temperature to 10oC.
A common (but imprecise) way of stating the third law of thermodynamics is "You can't reach absolute zero." Discuss how the third law, as stated in Section 3.2, puts limits on how low a temperature can be attained by various refrigeration techniques.
In a real turbine, the entropy of the steam will increase somewhat. How will this affect the percentages of liquid and gas at pointin the cycle? How will the efficiency be affected?
What do you think about this solution?
We value your feedback to improve our textbook solutions.