The ingenious Stirling engine is a true heat engine that absorbs heat from an external source. The working substance can be air or any other gas. The engine consists of two cylinders with pistons, one in thermal contact with each reservoir (see Figure 4.7). The pistons are connected to a crankshaft in a complicated way that we'll ignore and let the engineers worry about. Between the two cylinders is a passageway where the gas flows past a regenerator: a temporary heat reservoir, typically made of wire mesh, whose temperature varies
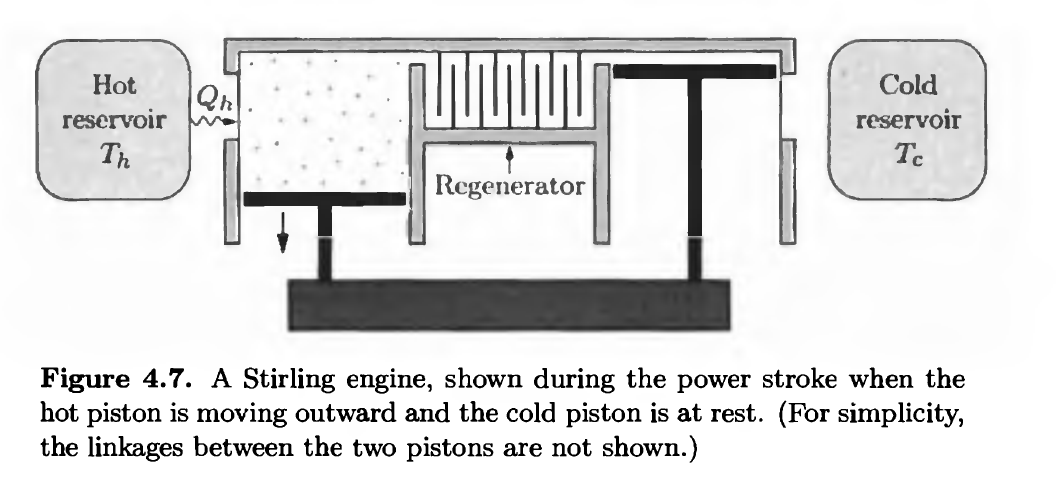
gradually from the hot side to the cold side. The heat capacity of the regenerator is very large, so its temperature is affected very little by the gas flowing past. The four steps of the engine's (idealized) cycle are as follows:
i. Power stroke. While in the hot cylinder at temperature Th, the gas absorbs heat and expands isothermally, pushing the hot piston outward. The piston in the cold cylinder remains at rest, all the way inward as shown in the figure.
ii. Transfer to the cold cylinder. The hot piston moves in while the cold piston moves out, transferring the gas to the cold cylinder at constant volume. While on its way, the gas flows past the regenerator, giving up heat and cooling to Tc.
iii. Compression stroke. The cold piston moves in, isothermally compressing the gas back to its original volume as the gas gives up heat to the cold reservoir. The hot piston remains at rest, all the way in.
iv. Transfer to hot cylinder. The cold piston moves the rest of the way in while the hot piston moves out, transferring the gas back to the hot cylinder at constant volume. While on its way, the gas flows past the regenerator, absorbing heat until it is again at Th.
(a) Draw a PV diagram for this idealized Stirling cycle.
(b) Forget about the regenerator for the moment. Then, during step 2, the gas will give up heat to the cold reservoir instead of to the regenerator; during step 4 , the gas will absorb heat from the hot reservoir. Calculate the efficiency of the engine in this case, assuming that the gas is ideal. Express your answer in terms of the temperature ratio Tc / Th and the compression ratio (the ratio of the maximum and minimum volumes). Show that the efficiency is less than that of a Carnot engine operating between the same temperatures. Work out a numerical example.
(c) Now put the regenerator back. Argue that, if it works perfectly, the efficiency of a Stirling engine is the same as that of a Carnot engine.
(d) Discuss, in some detail, the various advantages and disadvantages of a Stirling engine, compared to other engines.


