Chapter 1: Q. 1.69 (page 48)
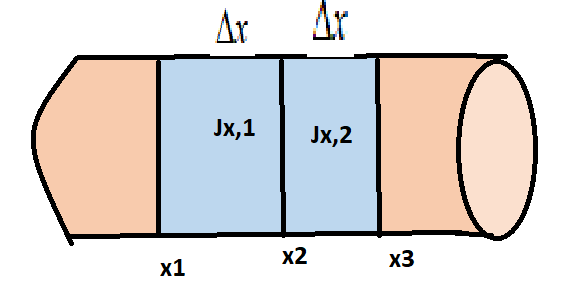
Consider a narrow pipe filled with fluid, where the concentration of a specific type of molecule varies only along its length (in the x direction). Fick's second law is derived by considering the flux of these particles from both directions into a short segment
Short Answer
Consider a narrow pipe filled with fluid, where the concentration of a specific type of molecule varies only along its length (in the x direction). The Fick's second law is