Chapter 0: Q11-39 P (page 1)
Write the chemical formula of a compound with a negative pK
Short Answer
The chemical formula of a compound with a negativehas to be written.
Learning Materials
Features
Discover
Chapter 0: Q11-39 P (page 1)
Write the chemical formula of a compound with a negative pK
The chemical formula of a compound with a negativehas to be written.
All the tools & learning materials you need for study success - in one app.
Get started for free
To pre-concentrate cocaine and benzoylecgonine from river water described at the opening of this chapter, solid-phase extraction was carried out at using the mixed-mode cation-exchange resin in Figure 28-19. After passing of river water through of resin, the retained analytes were eluted first with of and then with ammonia solution in
. Explain the purpose of using
for retention and dilute ammonia for elution.
Protein and carbohydrates provide, whereas fat gives . (Remember that 1 Calorie , with a capital C, is really .) The weight percentages of these components in some foods are
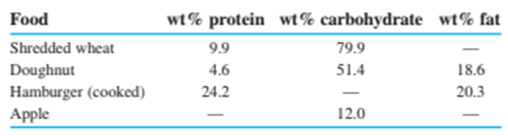
Calculate the number of calories per gram and calories per ounce in each of these foods. (Use Table 1-4to convert grams into ounces, remembering that there are 16ounces in 1pound.)
If there had beenreplications in both data sets, would the difference in standard deviations be significant?
The metal ion indicator xylenol orange (Table 12-3) is yellow at.The spectral changes that occur asis added to the indicator atpH6 are shown here. The mole ratio/xylenol orange at each point is
Suggest a sequence of chemical reactions to explain the spectral changes, especially the isosbestic points at 457 and 528nm.
Absorption spectra for the reaction of xylenol orange withatpH 6.0
How many grams of (FM 142.04) should be added to how many grams of sulfuric acid (FM 98.08) to give of buffer with pH 2.80 and a total sulfur concentration of ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.