Chapter 4: Q30P (page 93)
Explain the following statement: "The validity of a chemical analysis ultimately depends on measuring the response of the analytical procedure to known standards."
Short Answer
The given statement should be accepted.
Learning Materials
Features
Discover
Chapter 4: Q30P (page 93)
Explain the following statement: "The validity of a chemical analysis ultimately depends on measuring the response of the analytical procedure to known standards."
The given statement should be accepted.
All the tools & learning materials you need for study success - in one app.
Get started for free
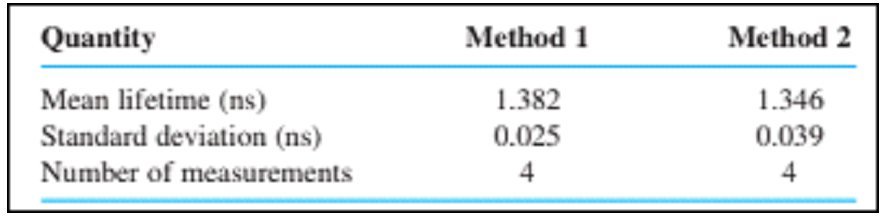
Two methods were used to measure fluorescence lifetime of a dye. Are the standard deviations significantly different? Are the means significantly different?
Spreadsheet for standard deviation. Let's create a spreadsheet to compute the mean and standard deviation of a column of numbers in two different ways. The spreadsheet here is a template for this exercise.
(a) Reproduce the template on your spreadsheet. Cells B4to B8contain the data ( xvalues) whose mean and standard deviation we will compute.
(b) Write a formula in cell B9to compute the sum of numbers in B4to B8.
(c) Write a formula in cell B10to compute the mean value.
(d) Write a formula in cell C4to compute (- mean), where xis in cellB4 and the mean is in cell B10. Use Fill Down to compute values in cells C5to C8.
(e) Write a formula in cellto compute the square of the value in cell. Use Fill Down to compute values in cellsto.
(f) Write a formula in cell D9 to compute the sum of the numbers in cells D4to D8.
(g) Write a formula in cell B11to compute the standard deviation.
(h) Use cells B13to B18to document your formulas.
(i) Now we are going to simplify life by using formulas built into the spreadsheet. In cell B21type which means find the sum of numbers in cells B4to B8. Cell B21should display the same number as cell B9. In general, you will not know what functions are available and how to write them. In Excel 2010, use the Formulas ribbon and Insert Function to find SUM.
(j) Select cellB22. Go to Insert Function and find AVERAGE. When you type "=AVERAGE(B4:B8)" in cell B22, its value should be the same asB10.
(k) For cellB23, find the standard deviation functionand check that the value agrees with cell B11.
If each of the four numbers 821,783,834,and855in the example is divided by 2, how will the mean, standard deviation, and coefficient of variation be affected?
Nonlinear calibration curve. Following the procedure in Box 4-2, find how many micrograms of protein are contained in a sample with a corrected absorbance of 0.350in Figure 4-13.
calibration curve based onknown points was used to measure the protein in an unknown. The results were protein , where the standard uncertainty is . Find the90%and 99%confidence intervals for protein in the unknown.
What do you think about this solution?
We value your feedback to improve our textbook solutions.