In 2002, workers at the Swedish National Food Administration discovered that heated, carbohydrate-rich foods, such as french fries, potato chips, and bread, contain alarming levels \((0.1to4\mu {\rm{g}}/{\rm{g}})\) of acrylamide, a known carcinogen\(36\).
After the discovery, simplified methods were developed to measure ppm levels of acrylamide in food. In one procedure,\(10\;{\rm{g}}\) of pulverized, frozen french fries were mixed for \(20\;{\rm{min}}\)with\(50\;{\rm{mL}}\) of \({{\rm{H}}_2}{\rm{O}}\)to extract acrylamide, which is very soluble in water \((216\;{\rm{g}}/100\;{\rm{mL}}).\)The liquid was decanted and centrifuged to remove suspended matter. The internal standard \(^2{{\rm{H}}_3}\)-acrylamide was added to\(1\;{\rm{mL}}\) of extract. A solid-phase extraction column containing \(100{\rm{mg}}\)of cation-exchange polymer with protonated sulfonic acid groups\(\left( { - {\rm{S}}{{\rm{O}}_3}{\rm{H}}} \right.)\) was washed twice with 1 -mL portions of methanol and twice with \(1 - {\rm{mL}}\)portions of water. The aqueous food extract \((1{\rm{mL}})\)was then passed through the column to bind protonated acrylamide \(\left( { - {\rm{NH}}_3^ + } \right)\)to sulfonate \(\left( { - {\rm{SO}}_3^ - } \right)on\)the column. The column was dried for\(30\;{\rm{s }}at\)\(0.3\)bar and then acrylamide was eluted with\(1\;{\rm{mL}}\) of \({{\rm{H}}_2}{\rm{O}}.\)Eluate was analyzed by liquid chromatography with a polar bonded phase. The chromatograms show the results moni- tored by ultraviolet absorbance or by mass spectrometry. The retention time of acrylamide is different on the two columns because they have different dimensions and different flow rates.
(a) What is the purpose of solid-phase extraction prior to chromatography? How does the ion-exchange sorbent retain acrylamide?
(b) Why are there many peaks when chromatography is monitored by ultraviolet absorbance?
(c) Mass spectral detection used selected reaction monitoring (Figure 22-33) with the \(m/z72 \to 55\)transition for acrylamide and \(75 \to 58fo{r^2}{{\rm{H}}_3}\)-acrylamide. Explain how this detection method works and suggest structures for the ions with \({\rm{m}}/{\rm{z}}72\)and 55 from acrylamide.
(d) Why does mass spectral detection give just one major peak?
(e) How is the internal standard used for quantitation with mass spectral detection?
(f) Where does \(^2{{\rm{H}}_3}\)-acrylamide appear with ultraviolet absorbance? With mass spectral selected reaction monitoring?
(g) Why does the mass spectral method give quantitative results even though retention of acrylamide by the ion-exchange sorbent is not quantitative and elution of acrylamide from the sorbent by \(1\;{\rm{mL}}\) of water might not be quantitative?
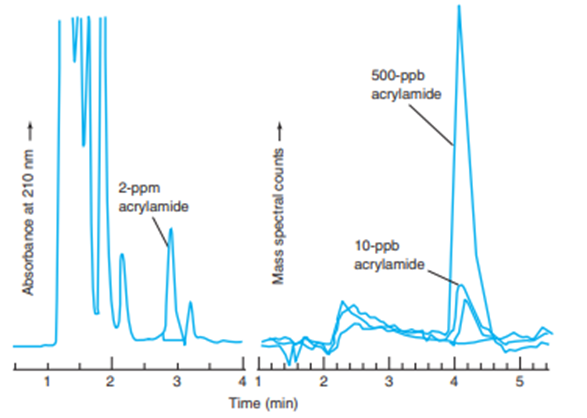
Chromatograms of acrylamide extract after passage through solid-phase extraction column. Left: Phenomenex Synergi Polar-RP 4- \(\mu {\rm{m}}\)column eluted with 96:4 \((vol/vol){{\rm{H}}_2}{\rm{O}}:{\rm{C}}{{\rm{H}}_3}{\rm{CN}}.\)Right: Phenomenex Synergi Hydro-RP 4- \(\mu {\rm{m}}\)column eluted with \(96:4:0.1(vol/vol/vol){{\rm{H}}_2}{\rm{O}}:{\rm{C}}{{\rm{H}}_3}{\rm{OH}}:{\rm{HC}}{{\rm{O}}_2}{\rm{H}}.\) (Data from L. Peng. T. Farkas, L. Loo, \({\rm{J}}.\)Teuscher, and \({\rm{K}}.\)Kallury, "Rapid and Reproducible Extraction of Acrylamide in French Fries Using a Single Solid-Phase Sorbent," Am. Lab. News Ed, October 2003, p. 10.)






