Chapter 9: Q5TY (page 199)
Find the pH if we add another 1.00g of tris hydrochloride.
Short Answer
The pH of buffered solution tris(hydroxymethyl)-aminomethane and tris hydrochloride is 8.53
Learning Materials
Features
Discover
Chapter 9: Q5TY (page 199)
Find the pH if we add another 1.00g of tris hydrochloride.
The pH of buffered solution tris(hydroxymethyl)-aminomethane and tris hydrochloride is 8.53
All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following acids would be most suitable for preparing a buffer of pH 3.10? (i) hydrogen peroxide; (ii) propanoic acid; (iii) cyanoacetic acid; (iv) 4-aminobenzenesulfonic acid.
Neglecting activity coefficients, calculate the pHof
Using activity coefficients correctly, find the pH of.
What concentration of OH- is produced by H2O dissociation in this solution? Was it justified to neglect water dissociation as a source of OH-?
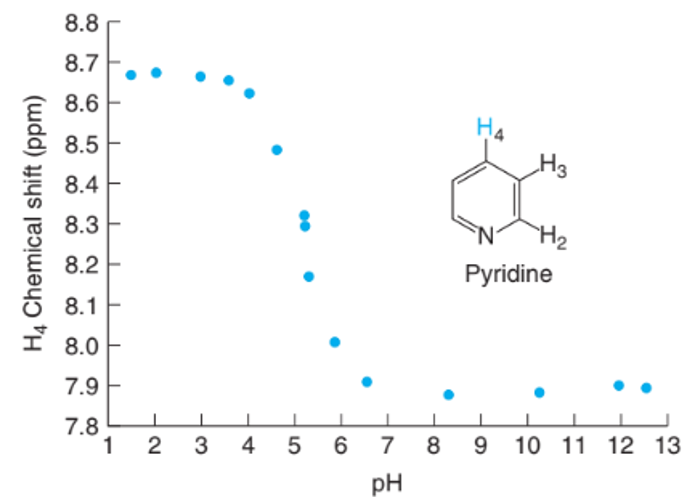
Interpreting spectral data. The graph shows the H-nuclear magnetic resonance chemical shift of the proton on pyridine as a function of Chemical shift is related to the environment of a proton in a molecule. If the environment changes, the chemical shift changes. Suggest an explanation for why the chemical shift changesbetween low and high Estimate for pyridinium ion
What do you think about this solution?
We value your feedback to improve our textbook solutions.