Chapter 9: Q1P (page 208)
Why doesn’t water produce , andwhen
is added?
Short Answer
HBr is a strong acid; when it is dissolved in water, it gets completely disassociated into and
Learning Materials
Features
Discover
Chapter 9: Q1P (page 208)
Why doesn’t water produce , andwhen
is added?
HBr is a strong acid; when it is dissolved in water, it gets completely disassociated into and
All the tools & learning materials you need for study success - in one app.
Get started for free
Would you needto bring the pH of HEPES (Table 9-2) to 7.45?
(b) Describe how to prepare HEPES, .
Describe how to prepare acetate buffer, ,starting with pure liquid acetic acid and solutions containing
How many mL of 0.500 M NaOH should be added to 10.0 g of tris hydrochloride to give a pH of 7.40 in a final volume of 500 mL?
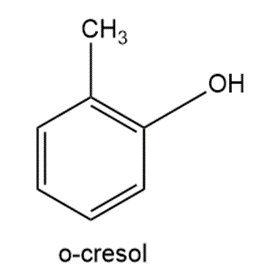
The pH of 0.010 M o-cresol is 6.16. Find pKa for this weak acid.
Find theand fraction of dissociation ( a ) of a solutionof the weak acid with
What do you think about this solution?
We value your feedback to improve our textbook solutions.