Chapter 23: Q40P (page 631)
Explain how slinization reduces tailing of chromatographic peaks.
Short Answer
Silanization creates hydroxyl groups that can form strong hydrogen bonds.
Learning Materials
Features
Discover
Chapter 23: Q40P (page 631)
Explain how slinization reduces tailing of chromatographic peaks.
Silanization creates hydroxyl groups that can form strong hydrogen bonds.
All the tools & learning materials you need for study success - in one app.
Get started for free
A chromatogram with ideal Gaussian bands hasand
.
(a) How many theoretical plates are present?
(b) Find the plate height if the column is 10cm long.
Which of the following columns will provide:
(a) highest number of plates?
(b) greatest retention?
(c) highest relative retention?
(d) best separation?
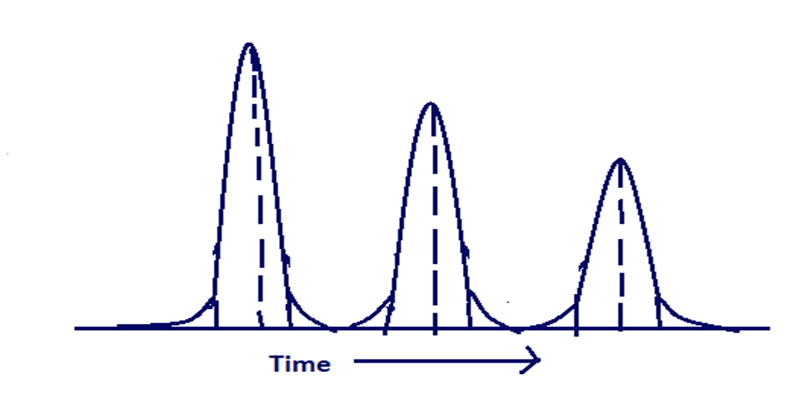
The three chromatograms shown here obtained with 2.5, 1.0 and 0.4 of ethyl acetate injected on the same column under the same conditions. Explain why the peak becomes less symmetrical with increasing sample size.
23-38. What is the optimal flow rate in Figure 23-17 for best separation of solutes?
Match statements 1–5 with the band broadening terms in the second list.
1. Depends on radius of open tubular column.
2. Not present in an open tubular column.
3. Depends on length and radius of connecting tubing.
4. Increases with diffusion coefficient of solute.
5. Increases with thickness of stationary phase film.
A Multiple paths
B Longitudinal diffusion
Equilibration time in mobile phase
Equilibration time in stationary phase
EC Extra column band broadening
What do you think about this solution?
We value your feedback to improve our textbook solutions.