23-32. Chromatograms of compounds A and B were obtained at the same flow rate with two columns of equal length. The value of is 1.3m in both cases.
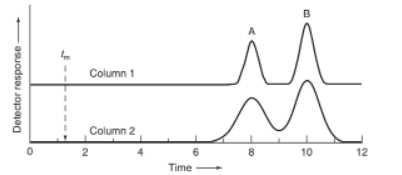
(a) Which column has more theoretical plates?
(b) Which column has a larger plate height?
(c) Which column gives higher resolution?
(d) Which column gives a greater relative retention?
(e) Which compound has a higher retention factor?
(f) Which compound has a greater partition coefficient?
(g) What is the numerical value of the retention factor of peak A?
(h) What is the numerical value of the retention factor of peak B?
(i) What is the numerical value of the relative retention?


