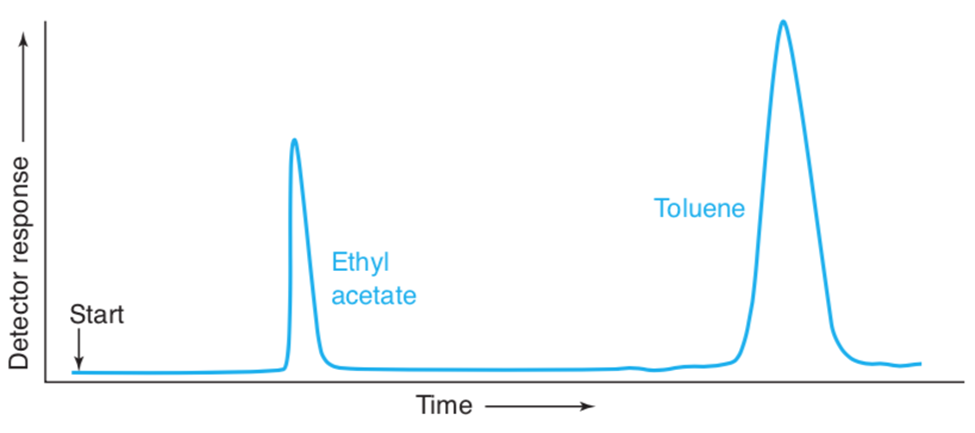
Chapter 23: Q16P (page 629)
The theoretical limit for extracting solute Sfrom into phase2 is attained by dividing into an infinite number of infinitesimally small portions and conducting an infinite number of extractions. With a partition coefficient, the limiting fraction of solute remaining in phase . Let volume be divided intoequal portions to conduct extractions. Find the fraction of S extracted into phase 2 for n = 1,2, and 10extractions. How many portions are required to attain 95%of the theoretical limit?
Short Answer
To conclude the number of portions required in order to attain the 95% of the limit would be and the extracted fraction would be