Chapter 24: Q4P. (page 663)
(a) What types of solutes are typically separated with a poly(dimethylsiloxane)-coated open tubular column?
(b) What types of solutes are typically separated with a poly(ethylene glycol)-coated open tubular column?
(c) What types of solutes are typically separated with a porous-layer open tubular column?
Short Answer
(a.)
Selecting a liquid stationary phase depends on the rule "like dissolves like". This signifies that the Polarity of the column must be the same as that of the solute
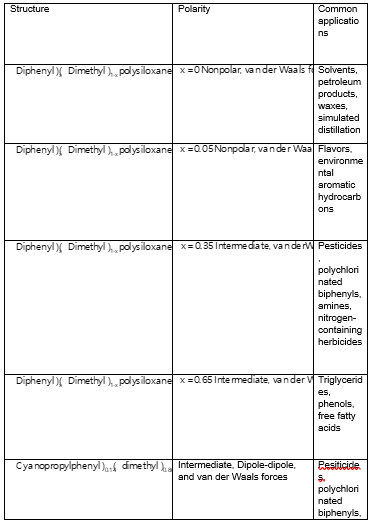
(b.) Solutes that are typically separated with a poly(dimethylsiloxane)-coated open tubular column are the following (based on Table 24-1)
(c.) Porous-layer columns contain porous solid particles with large surface area that adhere to the column wall. The highly retentive surface of the particles serve as the active stationary phase. Solutes that are typically separated with a porous-layer open tubular column . Furthermore, porous polymers, high-surface-area carbon , and alumina can separate hydrocarbons in gas-solid adsorption chromatography.