The antitumor drug gimatecan is available as nearly pure (S)-enantiomer. Neither pure (R)-enantiomer nor a racemic (equal) mixture of the two enantiomers is available. To measure small quantities of (R)-enantiomer in nearly pure (S)-gimatecan, a preparation was subjected to normal-phase chromatography on each of the enantiomers of a commercial, chiral stationary phase designated (S,S)- and (R,R)-DACH-DNB. Chromatography on the (R,R)-stationary phase gave a slightly asymmetric peak at tr 5 6.10 min with retention factor k 5 1.22. Chromatography on the (S,S)- stationary phase gave a slightly asymmetric peak at tr 5 6.96 min with k 5 1.50. With the (S,S) stationary phase, a small peak with 0.03% of the area of the main peak was observed at 6.10 min.
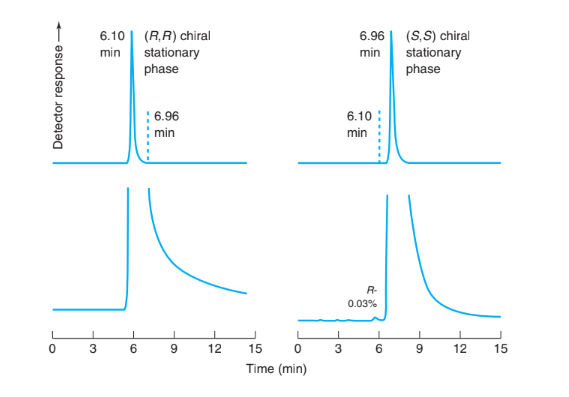
Chromatography of gimatecan on each enantiomer of a chiral stationary phase. Lower traces have enlarged vertical scale. [Data from E. Badaloni, W. Cabri, A. Ciogli, R. Deias, F. Gasparrini, F. Giorgi, A. Vigevani, and C. Villani, “Combination of HPLC ‘Inverted Chirality Columns Approach’ and MS/MS Detection for Extreme Enantiomeric Excess Determination Even in Absence of Reference Samples.” Anal. Chem. 2007, 79, 6013.]
(a) Explain the appearance of the upper chromatograms. Dashed lines are position markers, not part of the chromatogram. What Problems 709 would the chromatogram of pure (R)-gimatecan look like on the same two stationary phases?
(b) Explain the appearance of the two lower chromatograms and why it can be concluded that the gimatecan contained 0.03% of the (R)-enantiomer. Why is the (R)-enantiomer not observed with the (R,R)-stationary phase?
(c) Find the relative retention (a) for the two enantiomers on the (S,S)-stationary phase.
(d) The column provides N 5 6 800 plates. What would be the resolution between the two equal peaks in a racemic (equal) mixture of (R)- and (S)-gimatecan? If the peaks were symmetric, does this resolution provide baseline separation in which signal returns to baseline before the next peak begins?


