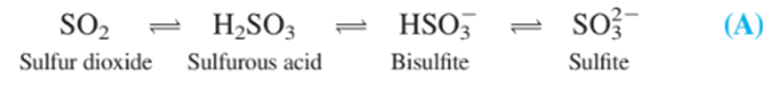Coulometric titration of sulfite in wine. Sulfur dioxide is added to many foods as a preservative. In aqueous solution, the following species are in equilibrium:
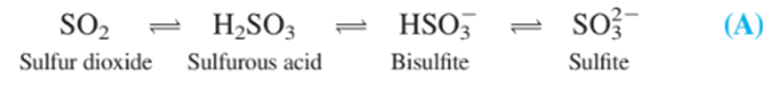
![]()
Bisulfite reacts with aldehydes in food near neutral pH:
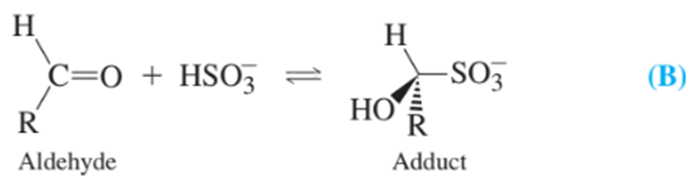
Sulfite is released from the adduct in 2MNaOH and can be analyzed by its reaction with to give and sulfate. Excess must be present for quantitative reaction.
Here is a coulometric procedure for analysis of total sulfite in white wine. Total sulfite means all species in Reaction and the adduct in Reaction . We use white wine so that we can see the color of a starch-iodine end point.
1. Mix 9.00 mL of wine plus 0.8gNaOH and dilute to 10.00mL. The releases sulfite from its organic adducts.
2. Generate at the working electrode (the anode) by passing a known current for a known time through the cell in Figure 17 - 10. The cell containsofacetate buffer () plus. In the cathode compartment, is reduced to . The frit retards diffusion of into the main compartment, where it would react with to give.
3. Generate at the anode with a current of for.
4. Inject 2.000mL of the wine/ solution into the cell, where the sulfite reacts with leaving excess.
5. Add of thiosulfate to consume by Reaction and leave excess thiosulfate.
6. Add starch indicator to the cell and generate fresh with a constant current of 10.0mA. A time of 131s was required to consume excess thiosulfate and reach the starch end point.
(a) In what pH range is each form of sulfurous acid predominant?
(b) Write balanced half-reactions for the anode and cathode.
(c) At pH 3.7, the dominant form of sulfurous acid isand the dominant form of sulfuric acid is . Write balanced reactions between andand between and thiosulfate.
(d) Find the concentration of total sulfite in undiluted wine.