Chapter 11: Q70P (page 263)
Titrating diprotic acid with strong base.
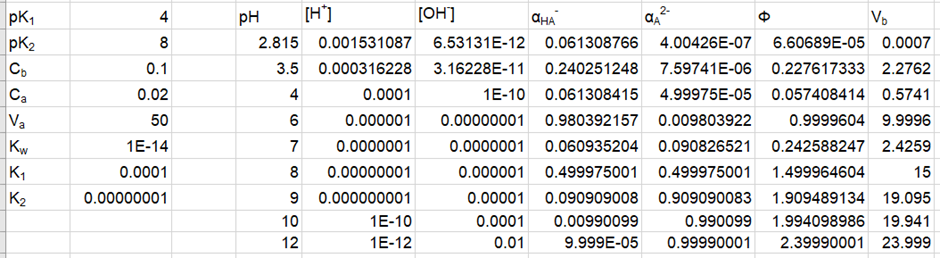
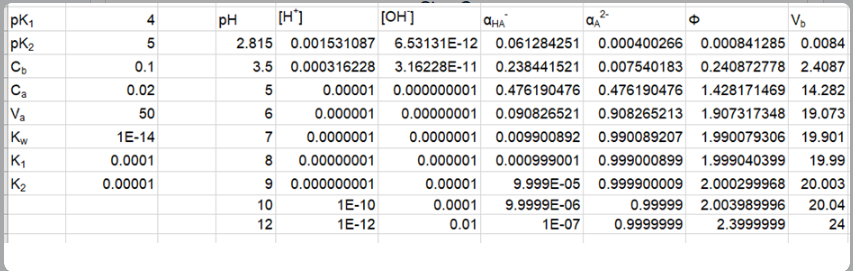
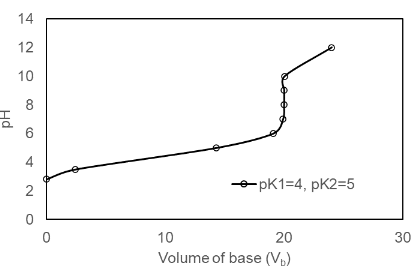
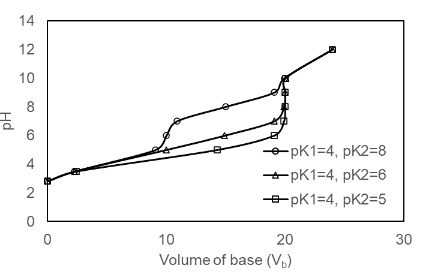
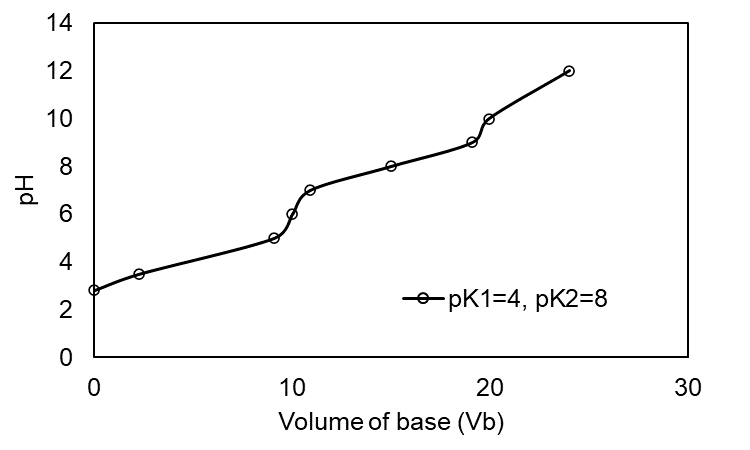
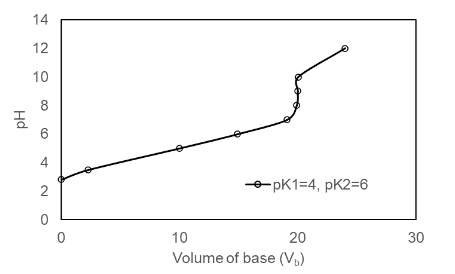
Prepare a family of graphs for the titration of 50.0 mL of 0.020 0 M H2A with 0.100 M NaOH. Consider the following cases: (a) pK1 = 4.00, pK2 =8.00; (b) pK1 = 4.00, pK2 = 6.00; (c) pK1 = 4.00, pK2 = 5.00
Short Answer
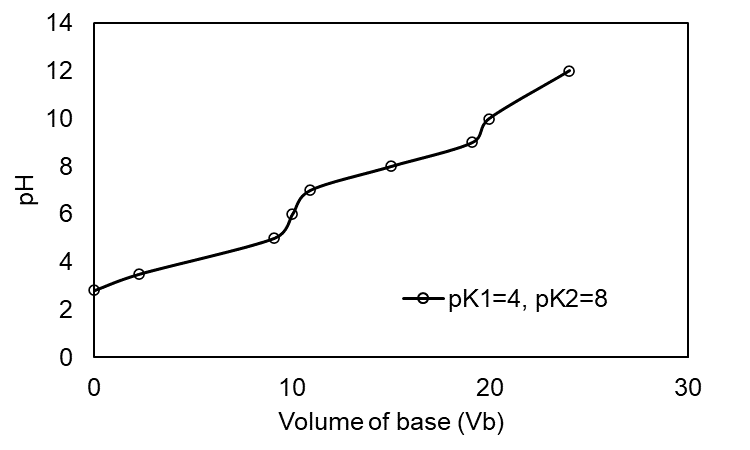
(a) The plot of the for pK1 = 4.00, pK2 =8.00 is shown below
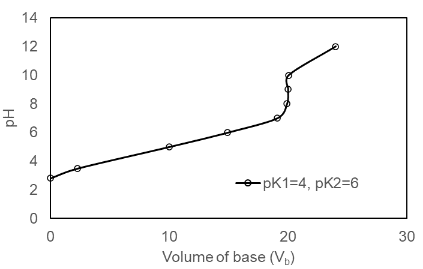
(b) The plot of the for pK1 = 4.00, pK2 =6.00 is shown below
(c) The plot of the for pK1 = 4.00, pK2 =5.00 is shown below