Chapter 11: Q29P (page 260)
Titration on Diprotic Systems
Find the pH of the solution when 0.0100M tyrosine is titrated to the equivalence point with0.00400MHCIO4.
Short Answer
The pH of the solution is 2.72.
Learning Materials
Features
Discover
Chapter 11: Q29P (page 260)
Titration on Diprotic Systems
Find the pH of the solution when 0.0100M tyrosine is titrated to the equivalence point with0.00400MHCIO4.
The pH of the solution is 2.72.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why does an acid-base titration curve (pH versus volume of titrant) have an abrupt change at the equivalence point?
Distinguish the terms end point and equivalence point.
Spectrophotometric properties of a particular indicator are given below:
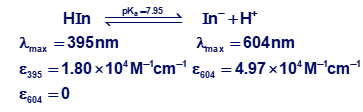
A solution with a volume of 20.0 mL containing 1.40 ×10-5 M indicator plus 0.050 0 M benzene-1,2,3-tricarboxylic acid was treated with 20.0 mL of aqueous KOH. The resulting solution had an absorbance at 604 nm of 0.118 in a 1.00-cm cell. Calculate the molarity of the KOH solution.
The Kjeldahl procedure was used to analyze 256 mL of a
solution containing 37.9 mg protein/mL The liberated NH3 was
collected in 5.00 mL of 0.033 6 M HCl, and the remaining acid
required 6.34 mL of 0.010 M NaOH for complete titration. What is
the weight percent of nitrogen in the protein?
Consider the titration of weak acid with . At what fraction of does ? At what fraction of does localid="1655007666473" ? Use these two points, plus to sketch the titration curve for the reaction of of localid="1655007634810" anilinium bromide (aminobenzene. ) withlocalid="1655007889912" .
What do you think about this solution?
We value your feedback to improve our textbook solutions.