Chapter 11: Q22 P (page 260)
11-22. The figure compares the titration of a monoprotic weak acid with a monoprotic weak base and the titration of a diprotic acid with a strong base.
(a) Write the reaction between the weak acid and the weak base and show that the equilibrium constant is . This large value means that the reaction goes "to completion" after each addition of the reagent.
(b) Why doesintersect the upper curve at and the lower curve at ? On the lower curve, is the acid, .
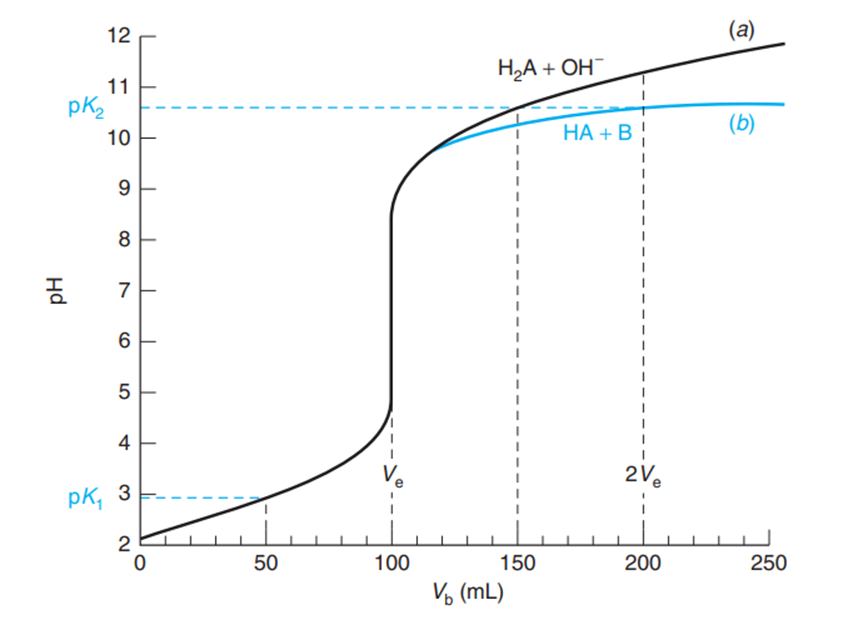
(a) Titration of 100mL of with 0.050 M NaOH.
(b) Titration of 100mL of the weak acid localid="1663575238403" with the weak baselocalid="1663575247899"


