Chapter 11: Q10P (page 233)
Whenof aqueouswere added toof cyclohexylaminoethanesulfonic acid structure in Table dissolved in of water, the pH was . Calculate the molarity of the
Short Answer
The molarity of is .
Learning Materials
Features
Discover
Chapter 11: Q10P (page 233)
Whenof aqueouswere added toof cyclohexylaminoethanesulfonic acid structure in Table dissolved in of water, the pH was . Calculate the molarity of the
The molarity of is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Finding the End Point with a pH Electrode
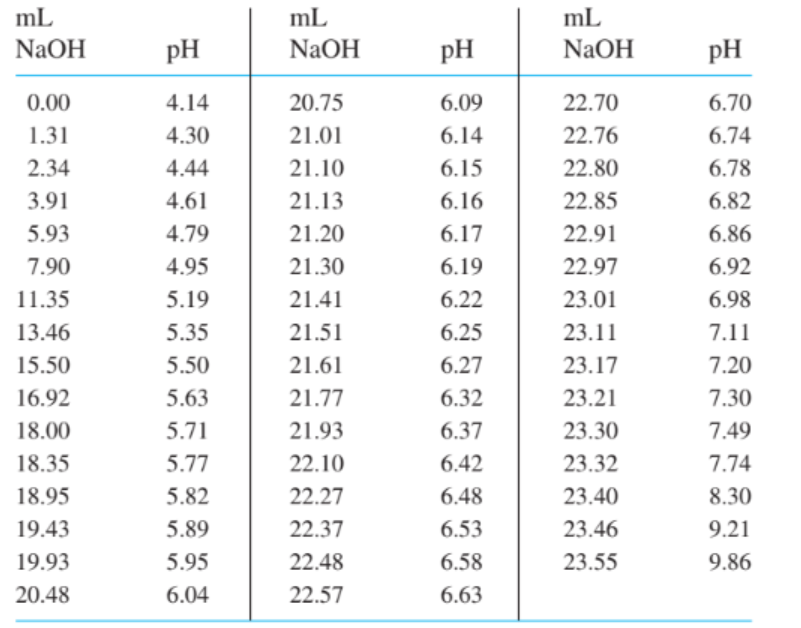
. Data for the titration of of a weak acid by are given below. Find the end point by preparing a Gran plot, using the last of the volume prior to.
solution of 0.0319Mbenzylamine was titrated with0.0500MHCl.Calculate thepHat the following volumes of added acid: , and 35.0mL.
Effect of pKb in the titration of weak base with strong acid.Using the appropriate equation in Table 11-5, compute and plot a family of curves analogous to the left part of Figure 11-3 for the titration of 50.0 mL of 0.020 0 M B (pKb = -2.00, 2.00, 4.00, 6.00, 8.00, and 10.00) with 0.100 M HCl. (The value pKb = -2.00 represents a strong base.) In the expression for
Titration on Diprotic Systems
. Calculate the whenrole="math" localid="1655019122863" of aminophenol have been titrated with role="math" localid="1655019100358" of .
Find the pH when Va = 14.63 mL.
What do you think about this solution?
We value your feedback to improve our textbook solutions.