Chapter 3: Q49 (page 190)
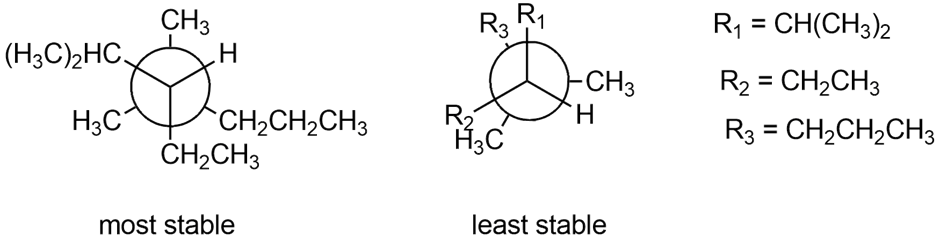
Question: Draw Newman projections along the C3-C4bond to show the most stable and least stable conformations of 3 ethyl-2,4demethylheptane.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 3: Q49 (page 190)
Question: Draw Newman projections along the C3-C4bond to show the most stable and least stable conformations of 3 ethyl-2,4demethylheptane.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Convert each Newman projection to the equivalent line-angle formula, and assign the IUPAC name.
Question: All of the following names are incorrect or incomplete. In each case, draw the structure ( or a possible structure) and name it correctly
Using what you know about the conformational energetics of substituted cyclohexanes, predict which of the two isomers is more stable. Explain your reasoning.
Name the following compounds. Remember the two up bonds are cis; two down bonds are cis; one up and one down bond are trans.
Table 3-6 shows that the axial-equatorial energy difference for methyl, ethyl, and isopropyl groups increases gradually: 7.6, 7.9 and 8.8 kJ/mol (1.8, 1.9, and 2.1 kcal/mol). The tert-butyl group jumps to an energy difference of 23 kJ/mol (5.4 kcal/mol), over twice the value for the isopropyl group. Draw pictures of the axial conformations of isopropylcyclohexane and tert-butylcyclohexane and explain why the tert-butyl substituent experiences such a large increase in axial energy over the isopropyl group.
What do you think about this solution?
We value your feedback to improve our textbook solutions.