Chapter 3: Q22P (page 176)
Draw 1,2,3,4,5,6-hexamethylcyclohexane with all the methyl groups
(a) in axial positions. (b) in equatorial positions.
Short Answer
(a)
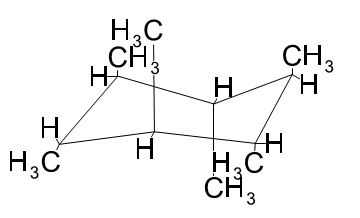
(b)
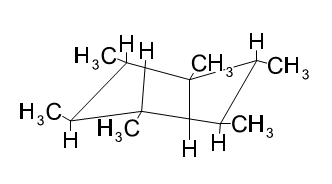
Learning Materials
Features
Discover
Chapter 3: Q22P (page 176)
Draw 1,2,3,4,5,6-hexamethylcyclohexane with all the methyl groups
(a) in axial positions. (b) in equatorial positions.
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Write structures for a homologous series of alkyl chlorides having from one to six carbons.
Table 3-6 shows that the axial-equatorial energy difference for methyl, ethyl, and isopropyl groups increases gradually: 7.6, 7.9 and 8.8 kJ/mol (1.8, 1.9, and 2.1 kcal/mol). The tert-butyl group jumps to an energy difference of 23 kJ/mol (5.4 kcal/mol), over twice the value for the isopropyl group. Draw pictures of the axial conformations of isopropylcyclohexane and tert-butylcyclohexane and explain why the tert-butyl substituent experiences such a large increase in axial energy over the isopropyl group.
(a) Draw both chair conformations of cis - 1,4 -dimethylcyclohexane and determine which conformer is more stable.
(b) Repeat for the trans isomer.
(c) Predict which isomer (cis or trans) is more stable.
Using what you know about the conformational energetics of substituted cyclohexanes, predict which of the two isomers is more stable. Explain your reasoning.
What do you think about this solution?
We value your feedback to improve our textbook solutions.