Chapter 3: Q15P (page 167)
Give IUPAC names for the following compounds.
(a)
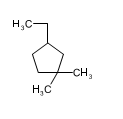
(b)
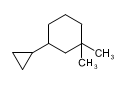
(c)
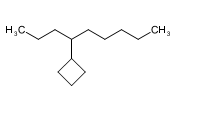
Short Answer
(a)3-ethyl-1,1-dimethyl cyclopentane
(b)3-cyclopropyl-1,1-dimethylcyclohexane
(c)nonan-4-ylcyclobutane
Learning Materials
Features
Discover
Chapter 3: Q15P (page 167)
Give IUPAC names for the following compounds.
(a)

(b)

(c)

(a)3-ethyl-1,1-dimethyl cyclopentane
(b)3-cyclopropyl-1,1-dimethylcyclohexane
(c)nonan-4-ylcyclobutane
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: This is a Newman projection of a substituted cyclohexane.
(a) Draw the equivalent chair form
(b)Draw the equivalent structure using wedge and dash notation on a cyclohexane hexagon.
(c) Give the IUPAC name.
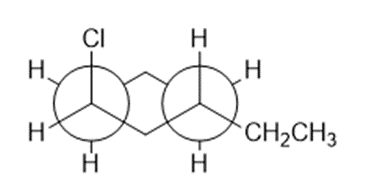
Draw the structure that corresponds with each name.
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
(i)
(j)
(k)
(l)
There are eight different five-carbon alkyl groups.
(a) Draw them
(b) Give them systematic names.
(c) In each case, label the degree of substitution (primary, secondary, or tertiary) of the head carbon atom bonded to the main chain.
The cyclohexane chair shown in Figure 3-22 has the headrest to the right and the footrest to the left. Draw a cyclohexane chair with its axial and equatorial bonds, showing the headrest to the left and the footrest to the right.
Draw the structure and give the molecular formula for each of the following compounds.
(a) 1-ethyl-3-methylcycloheptane
(b) isobutylcyclohexane
(c) cyclopropylcyclopentane
(d) 3-ethyl-1,1-dimethylcyclohexane
(e) 3-ethyl-2,4-dimethylhexane
(f) 1,1-diethyl-4-(3,3-dimethylbutyl)cyclohexane
What do you think about this solution?
We value your feedback to improve our textbook solutions.