Chapter 3: 28P (page 181)
Use your results from Problem 3-27 to complete the following table. Each entry shows the positions of two groups arranged as shown. For example, two groups that are trans on adjacent carbons must be equatorialor both axial.
Learning Materials
Features
Discover
Chapter 3: 28P (page 181)
Use your results from Problem 3-27 to complete the following table. Each entry shows the positions of two groups arranged as shown. For example, two groups that are trans on adjacent carbons must be equatorialor both axial.
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the most stable conformation of
(a) ethylcyclohexane
(b) 3-isopropyl-1,1-dimethylcyclohexane
(c) cis - 1-tert- butyl - 4 - isopropylcyclohexane
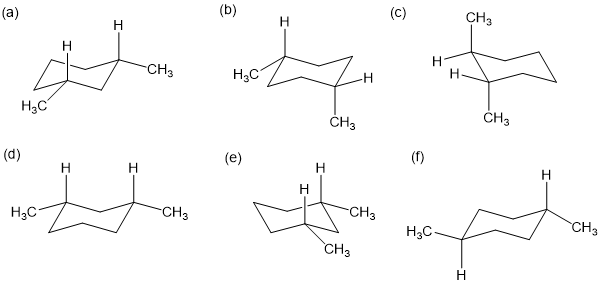
Name the following compounds. Remember the two up bonds are cis; two down bonds are cis; one up and one down bond are trans.
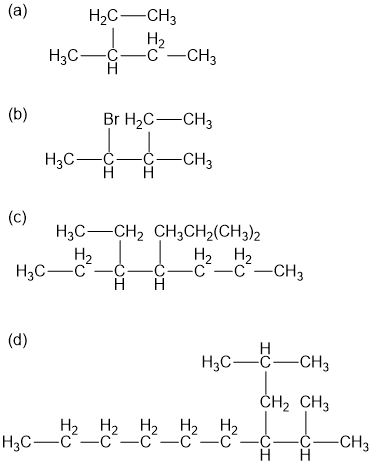
Name the following alkane and haloalkane. When two or more are present list them in alphabetical order.
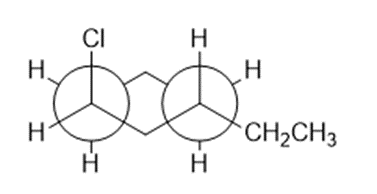
Question: This is a Newman projection of a substituted cyclohexane.
(a) Draw the equivalent chair form
(b)Draw the equivalent structure using wedge and dash notation on a cyclohexane hexagon.
(c) Give the IUPAC name.
Draw the both chair conformations of each of the following substituted cyclohexanes. In each case, label the more stable conformation.
(a)
(b)
(c)
(d)
What do you think about this solution?
We value your feedback to improve our textbook solutions.