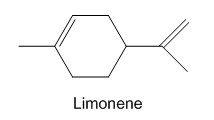
Limonene is one of the compounds that give lemons their tangy odour.Show the structures of the products expected when limonene reacts with an excess of each of these reagents.
(a) Borane in tetrahydrofuran, followed by basic hydrogen peroxide
(b) m-chloroperoxybenzoic acid
(c) ozone, then dimethyl sulfide
(d) a mixture of osmic acid and hydrogen peroxide
(e) hot, concentrated potassium permanganate
(f) peroxyacetic acid in acidic water
(g) hydrogen and a platinum catalyst
(h) hydrogen bromide gas
(i) hydrogen bromide gas in a solution containing dimethyl peroxide
(j) bromine water
(k) chlorine gas
(l) mercuric acetate in methanol, followed by sodium borohydride
(m) CHBr3 and 50% aq. NaOH


 Formation of iodonium ion
Formation of iodonium ion Intramolecular attack of carbonyl oxygen as nucleophile
Intramolecular attack of carbonyl oxygen as nucleophile

