Chapter 8: Q15P (page 418)
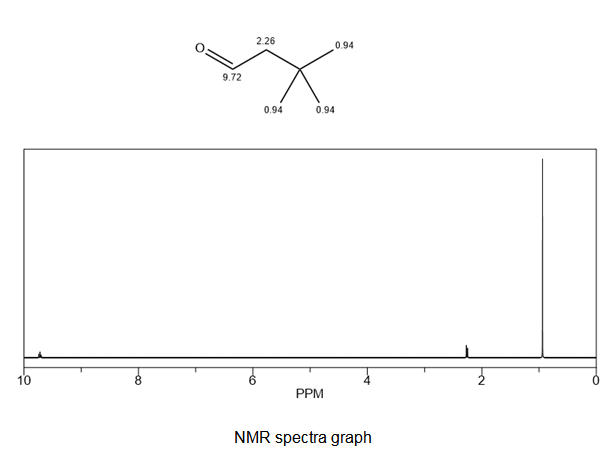
Sketch the expected proton NMR spectra of 3,3- dimethylbutanal.
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q15P (page 418)
Sketch the expected proton NMR spectra of 3,3- dimethylbutanal.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show how you would accomplish the following conversions.
(a)Cis-hex-3-ene to meso-hexane-3,4-diol
(b)Cis-hex-3-ene to (d,l)-hexane-3,4-diol
(c)Trans-hex-3-ene to meso-hexane-3,4-diol
(d)Trans-hex-3-ene to (d,l)-hexane-3,4-diol
Predict the major products of the following reactions.
(a) Propene + BH3 .THF (b) the product from part (a) + H2O2/OH-
(c) 2-methylpent-2-ene + BH3.THF (d) the product from part (c) + H2O2/OH-
(e) methylcyclohexane+ BH3 .THF (f) the product from (e) + H2O2/OH-
(a) When HBr adds across the double bond of 1,2-dimethylcyclopentene, the product is a mixture of the cis and trans isomers. Show why this addition is not stereospecific.
(b) When 1,2 dimethylcyclopropene undergoes hydroboration oxidation, one diastereomer of the product predominantes. Show why this addition is stereospecific, and predict the stereochemistry of the major product.
Predictthe major products of the following reactions.
a. E- 3-methyloct-3-ene+ ozone, then Me3S
b. Z- 3-methyloct-3-ene+ ozone, then Me3S
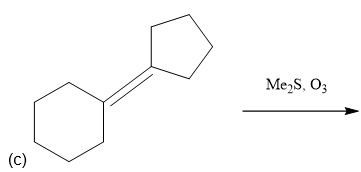
e. 1-ethylcycloheptene + ozone, then Me2S
f. 1-ethylcycloheptene + warm, then KMnO4
g. 1-ethylcycloheptene + cold,, dil.KMnO4
Predict the carbenoid addition products of the following reactions.
(a) trans-hex-3-ene+ CH2I2, Zn(Cu)
(b) ciz-hept-2-ene+ CH2I2, Zn(Cu)
What do you think about this solution?
We value your feedback to improve our textbook solutions.