Chapter 8: Q11P. (page 415)
Show how you would accomplish the following synthetic conversions.
(a)But-1-ene -> butan-1-ol
(b)But-1-ene ->butan-2-ol
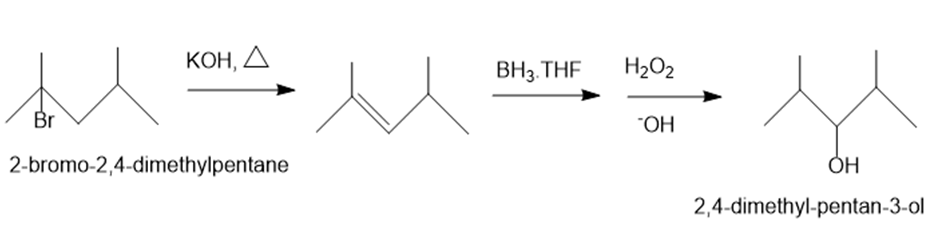
(c)2-bromo-2,4-dimethylpentane ->2,4-dimethylpentan-3-ol
Short Answer
(a)
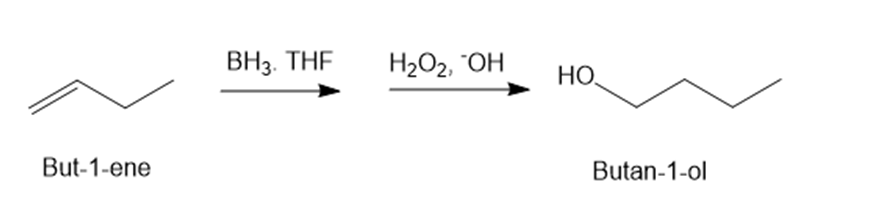
(b)
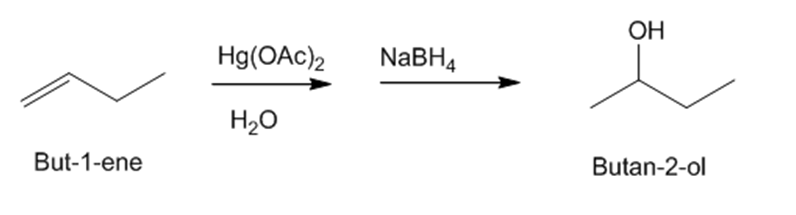
(c)
Learning Materials
Features
Discover
Chapter 8: Q11P. (page 415)
Show how you would accomplish the following synthetic conversions.
(a)But-1-ene -> butan-1-ol
(b)But-1-ene ->butan-2-ol
(c)2-bromo-2,4-dimethylpentane ->2,4-dimethylpentan-3-ol
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would accomplish each of the following synthetic conversions.
a) Trans-but-2-enetrans-1,2-dimethylcyclopropane
b) Cyclopentene
c) Cyclohexanol
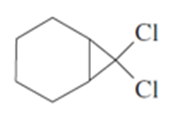
Predict the carbenoid addition products of the following reactions.
(a) trans - hex - 3 - ene + CH2I2, Zn(Cu)
(b) cis - hept - 2 - ene + CH2I2, Zn(Cu)
Question: Show how you would accomplish the following synthetic conversions.
Limonene is one of the compounds that give lemons their tangy odour.Show the structures of the products expected when limonene reacts with an excess of each of these reagents.
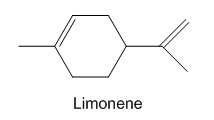
(a) Borane in tetrahydrofuran, followed by basic hydrogen peroxide
(b) m-chloroperoxybenzoic acid
(c) ozone, then dimethyl sulfide
(d) a mixture of osmic acid and hydrogen peroxide
(e) hot, concentrated potassium permanganate
(f) peroxyacetic acid in acidic water
(g) hydrogen and a platinum catalyst
(h) hydrogen bromide gas
(i) hydrogen bromide gas in a solution containing dimethyl peroxide
(j) bromine water
(k) chlorine gas
(l) mercuric acetate in methanol, followed by sodium borohydride
(m) CHBr3 and 50% aq. NaOH
The structures of three monomers are shown. In each case, show the structure of the polymer that would result from polymerization of the monomer. Vinyl chloride is polymerized to ‘’vinyl’’ plastics and PVC pipe. Tetrafluoroethylene polymerizes to Teflon, used as non-stick coatings and PTFE valves and gaskets. Acrylonitrile is polymerized to Orlon, used in sweaters and carpets.
What do you think about this solution?
We value your feedback to improve our textbook solutions.