Chapter 8: 28Q (page 429)
Show how you would accomplish each of the following synthetic conversions.
Short Answer

Learning Materials
Features
Discover
Chapter 8: 28Q (page 429)
Show how you would accomplish each of the following synthetic conversions.


All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the carbenoid addition products of the following reactions.
(a) trans - hex - 3 - ene + CH2I2, Zn(Cu)
(b) cis - hept - 2 - ene + CH2I2, Zn(Cu)
One of the principal components of lemongrass oil is limonene, . When limonene is treated with excess hydrogen and a platinum catalyst, the product is an alkane of formula . What can you conclude about the structure of limonene?
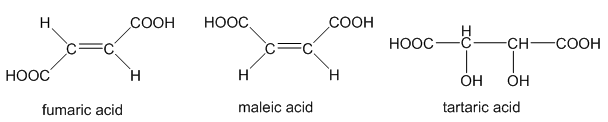
The two butenedioic acids are called fumaric acid(trans) and maleic acid(cis). 2,3-Dihydroxybutanedioic acid is called tartaric acid.
Show how would you convert
Predictthe major products of the following reactions.
a. E- 3-methyloct-3-ene+ ozone, then Me3S
b. Z- 3-methyloct-3-ene+ ozone, then Me3S
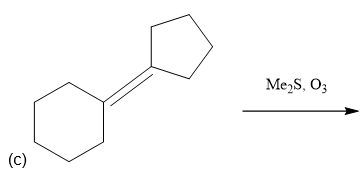
e. 1-ethylcycloheptene + ozone, then Me2S
f. 1-ethylcycloheptene + warm, then KMnO4
g. 1-ethylcycloheptene + cold,, dil.KMnO4
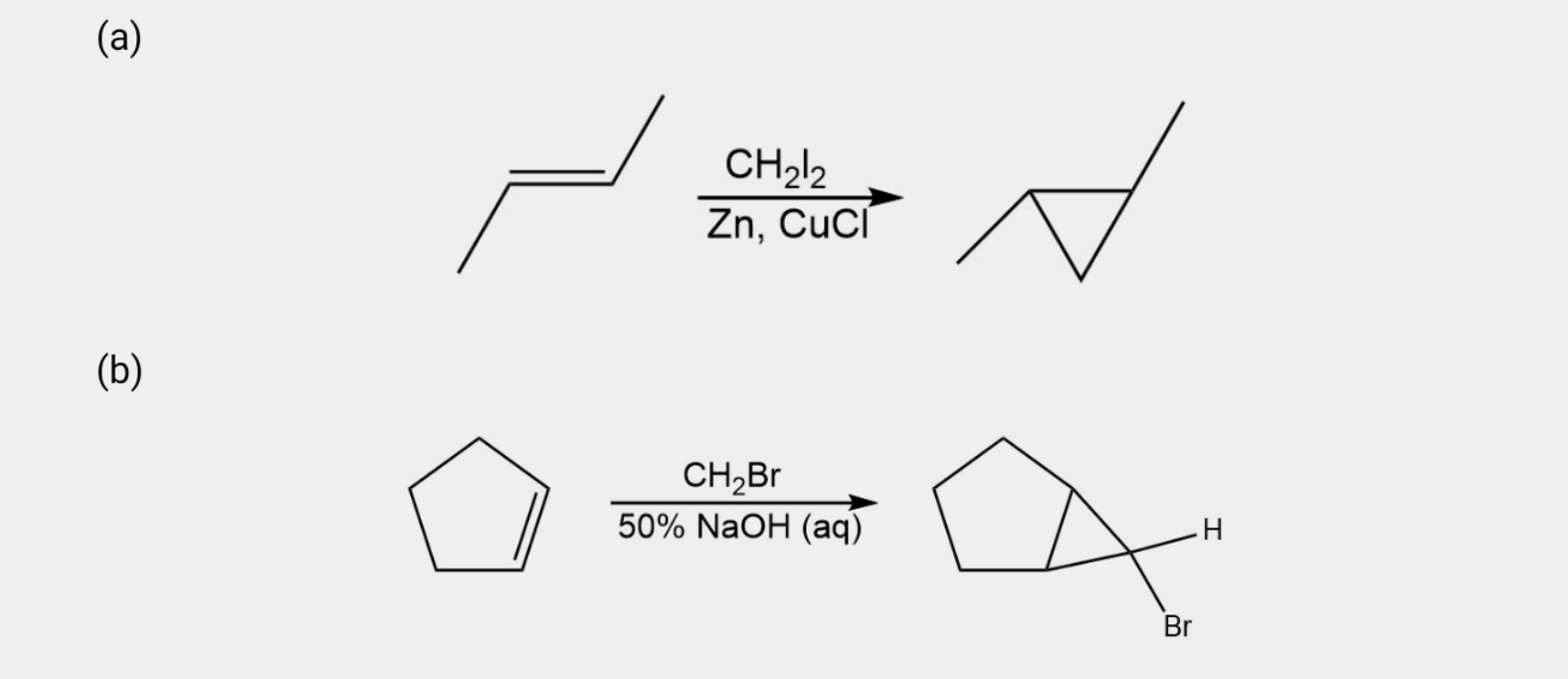
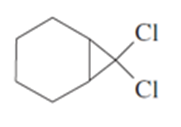
Show how you would accomplish each of the following synthetic conversions.
a) Trans-but-2-enetrans-1,2-dimethylcyclopropane
b) Cyclopentene
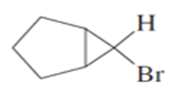
c) Cyclohexanol
What do you think about this solution?
We value your feedback to improve our textbook solutions.