Chapter 13: Q18P (page 675)
Predict the theoretical number of different NMR signals produced by each compound and give approximate chemical shifts. Point out any diastereotopic relationships.
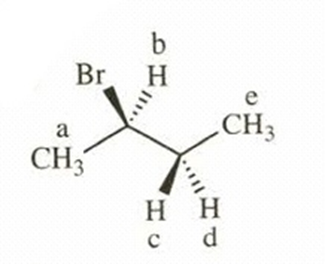
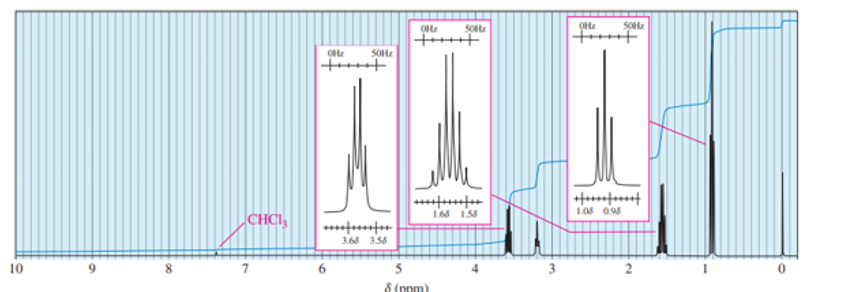
- 2-bromobutane
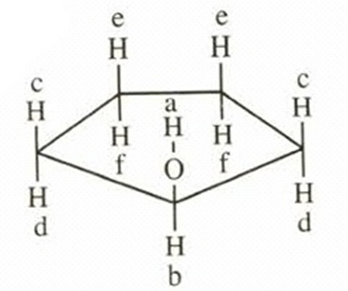
- Cyclopentanol
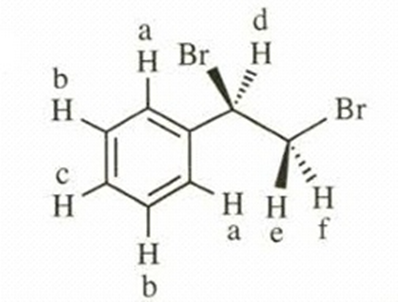
- Ph-CHBr-CH2Br
- Vinyl chloride
Short Answer
This compound shows five NMR signals. HCand HDare diastereotopic. a = δ 1.5, b = δ 3.6, c, d = δ 1.7, e = δ 1.0
This compound shows six NMR signals. HC and HD are diastereotopic. Also, HE and HF are diastereotopic.a = δ 2.5, b = δ 3.9, c, d = δ 1.6, e, f = δ 1.3
This compound shows six NMR signals. HE and HF are diastereotopic. a, b, c = δ 7.2, d = δ 5.0, e, f = δ 3.6
This compound shows three NMR signals. HA and HB are diastereotopic. a, b = δ 5-6, c = δ 7-8