Chapter 25: Q3P (page 1305)
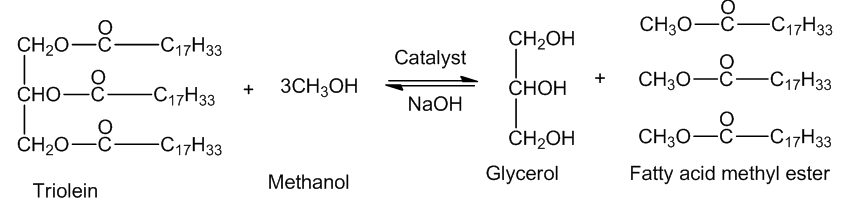
Give an equation for the complete transesterification of triolein using an excess of methanol as the alcohol and sodium hydroxide as the catalyst.
Short Answer
Learning Materials
Features
Discover
Chapter 25: Q3P (page 1305)
Give an equation for the complete transesterification of triolein using an excess of methanol as the alcohol and sodium hydroxide as the catalyst.

All the tools & learning materials you need for study success - in one app.
Get started for free
A triglyceride can be optically active if it contains two or more different fatty acids
The long-term health effects of eating partially hydrogenated vegetable oils concern some nutritionists because many unnatural fatty acids are produced. Consider the partial hydrogenation of linolenic acid by the addition of one or two equivalents of hydrogen. Show how this partial hydrogenation can produce at least three different fatty acids we have not seen before.
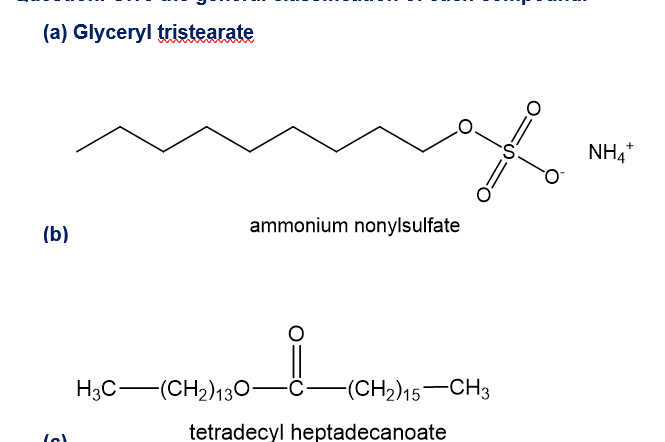
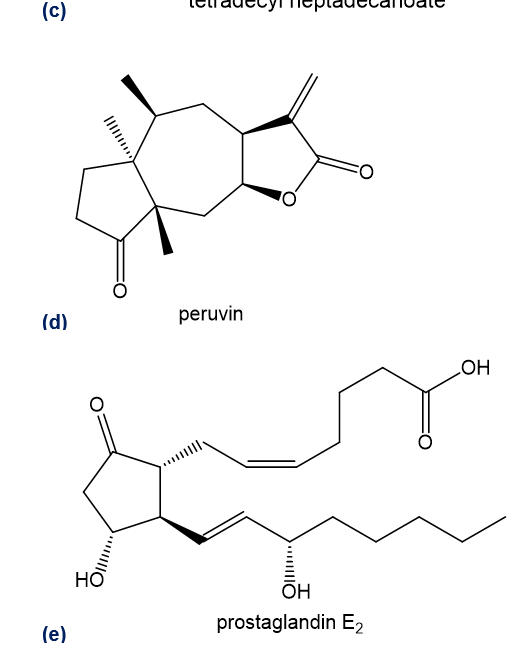
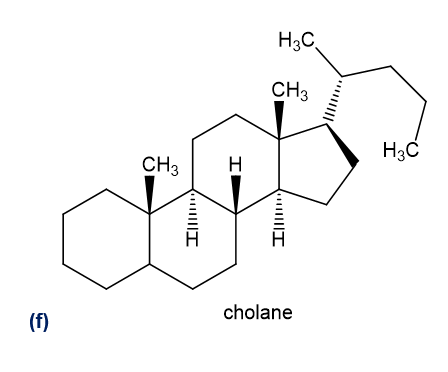
Give the general classification of each compound.
Show how you would convert linoleic acid to the following fatty acid derivatives.
Phospholipids undergo saponification much like triglycerides. Draw the structure of a phospholipid meeting the following criteria. Then draw the products that would result from its saponification.
(a) a cephalin containing stearic acid and oleic acid
(b) a lecithin containing palmitic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.