Chapter 18: Q66P (page 971)
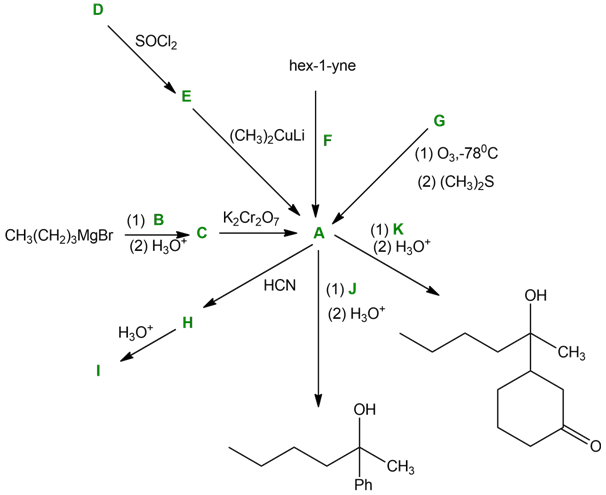
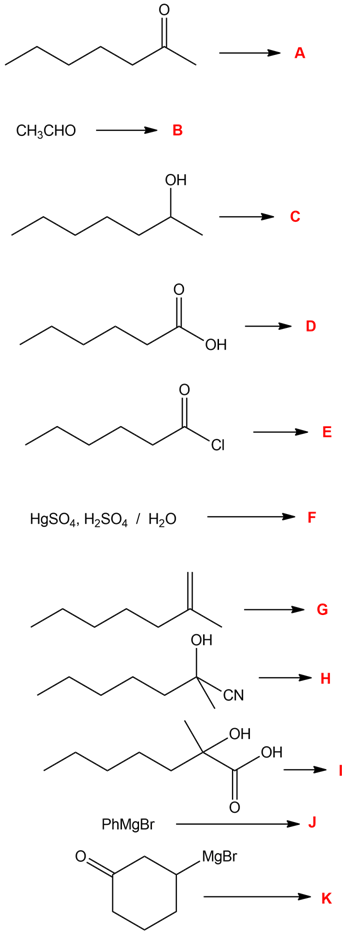
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q66P (page 971)
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K.


All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following compounds would give a positive Tollens test? (Remember that the Tollens test involves mild basic aqueous conditions)
(a) CH3CH2COCH3
(b) CH3CH2CHO
(c) CH3CH2CH=CHCH=CHOH
(d) CH3CH2 CH2CH(OH)OCH3
(e) CH3CH2 CH2CH(OCH3)2
(f)
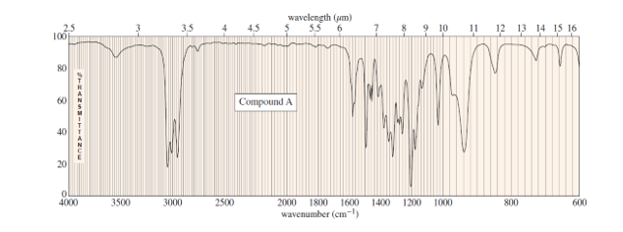
Question. The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow. When A is washed with dilute aqueous acid, extracted into dichloromethane , and the solvent evaporated, it gives a product B. B shows a strong carbonyl signal at 1715 cm-1in the IR spectrum and a weak maximum at 274nm(E =16) in the UV spectrum. The mass spectrum of B shows a molecular ion of m/z 72. Determine the structures of A and B, and show the fragmentation to account for the peaks at m/z 87 and 101.
PROBLEM 18-5
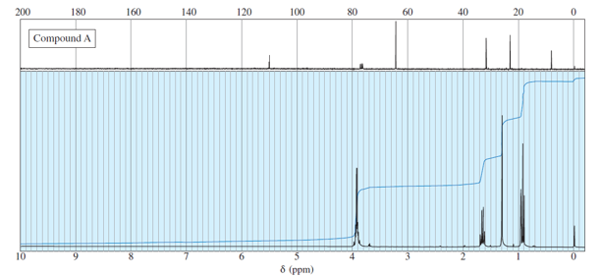
Oxidation of cholesterol converts the alcohol to a ketone. Under acidic or basic oxidationconditions, the C=C double bond migrates to the more stable, conjugated position. BeforeIR and NMR spectroscopy, chemists watched the UV spectrum of the reaction mixture tofollow the oxidation. Describe how the UV spectrum of the conjugated product, cholest-4-en-3-one, differs from that of cholesterol.
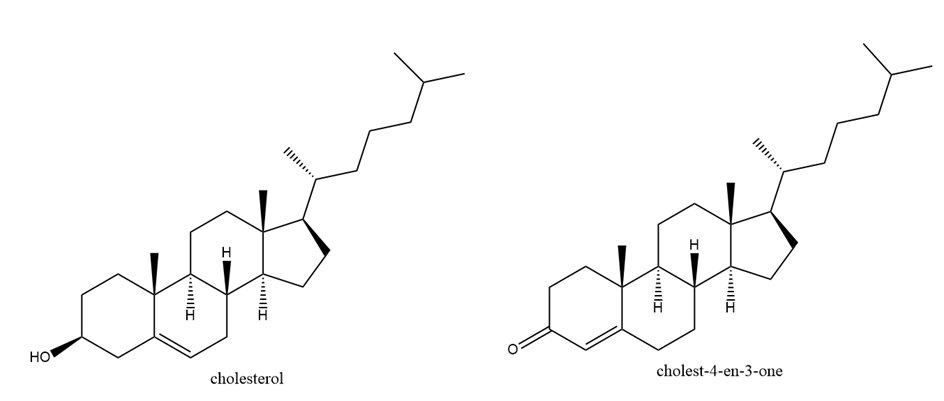
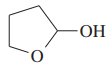
One of these reacts with dilute aqueous acid and the other does not. Give a mechanism for the one that reacts and show why this mechanism does not work for the other one.
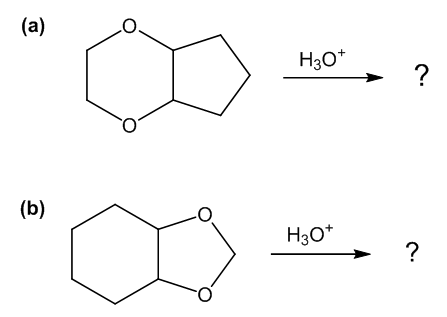
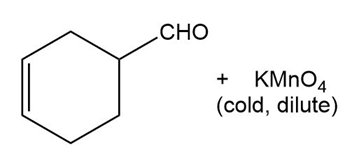
Predict the major products of the following reactions.
(a) (b)

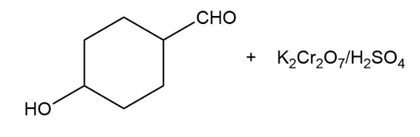
(c) (d)

What do you think about this solution?
We value your feedback to improve our textbook solutions.