Chapter 22: Q82P (page 1207)
Propose a mechanism for the following reaction. Show the structure of the compound that results from hydrolysis and decarboxylation of the product.
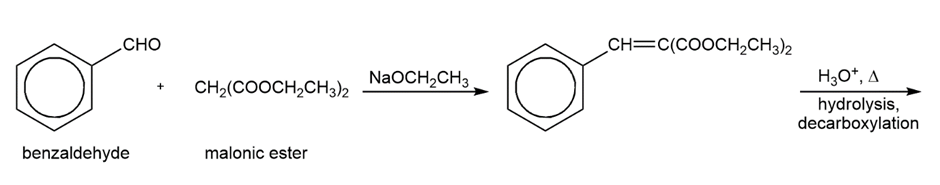
Short Answer
 Reaction Mechanism
Reaction Mechanism
Learning Materials
Features
Discover
Chapter 22: Q82P (page 1207)
Propose a mechanism for the following reaction. Show the structure of the compound that results from hydrolysis and decarboxylation of the product.

 Reaction Mechanism
Reaction Mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
When propionaldehyde is warmed with sodium hydroxide, one of the products is 2-methylpent-2-enal. Propose a mechanism for this reaction.
For each molecule shown below,
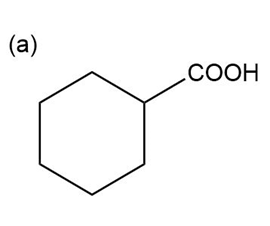

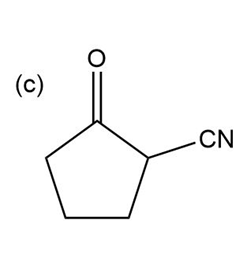
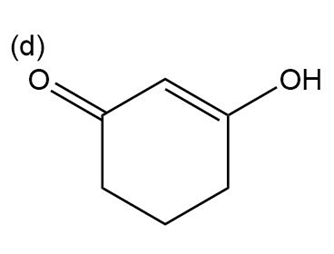
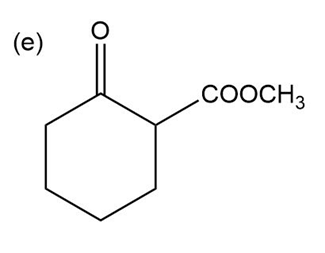
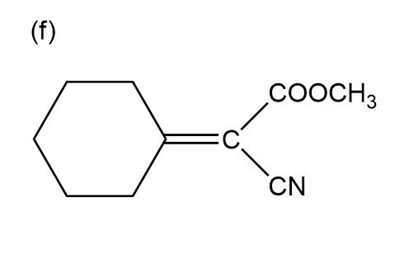
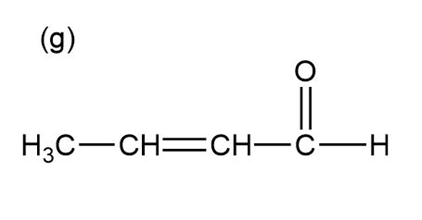
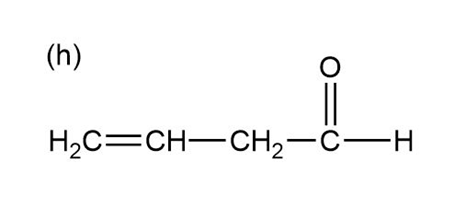
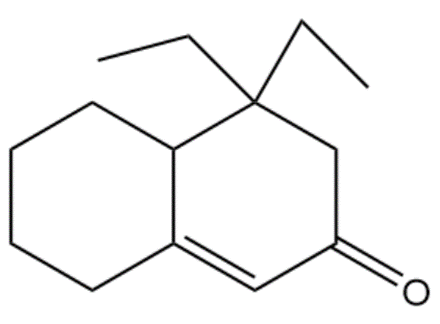
Question:Show how you would use the Robinson annulation to synthesize the following compounds.
A student wanted to dry some diacetone alcohol and allowed it to stand over anhydrous potassium carbonate for a week. At the end of the week, the sample was found to contain nearly pure acetone. Propose a mechanism for the reaction that took place.
Show how you would use the acetoacetic ester synthesis to make the following compounds

What do you think about this solution?
We value your feedback to improve our textbook solutions.